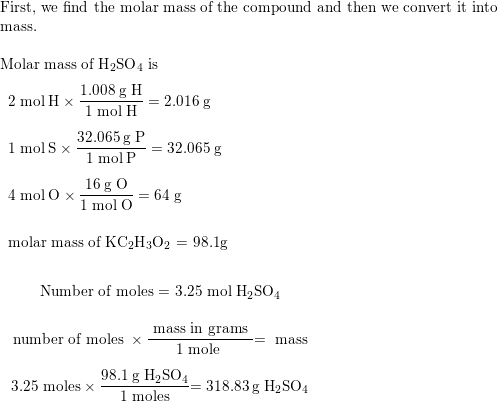SOLVED: 4. What is the molar mass of 3.25 mol of H2SO4 Points) Enter your answer 5. What is the molar mass of 2.5 mol of (C3H5)2 ? Points) Enter your answer
![Calculate the mass percentage composition of the elements in sulphuricr acid. [(H = 1, O = 16, S = 32)Atomic mass] Calculate the mass percentage composition of the elements in sulphuricr acid. [(H = 1, O = 16, S = 32)Atomic mass]](https://dwes9vv9u0550.cloudfront.net/images/1829042/c6d30049-457a-4243-bd97-a1c6b7154bcd.jpg)
Calculate the mass percentage composition of the elements in sulphuricr acid. [(H = 1, O = 16, S = 32)Atomic mass]

SOLVED: 9.8 g of H2SO4 is dissolved in enough water to make 500 mL of solution. The molarity of H+ and So ion in the solution respectively are (Assume 100% ionisation of

molecular mass/molar mass/molecular weight for sulfuric acid (H₂SO₄) , by khushboo yadav #molarmass - YouTube

Campbell / Callis C142B) Chapter #3 : Stoichiometry -Mole - Mass Relationships in Chemical Systems 3.1: The Mole 3.2: Determining the Formula of an Unknown. - ppt download
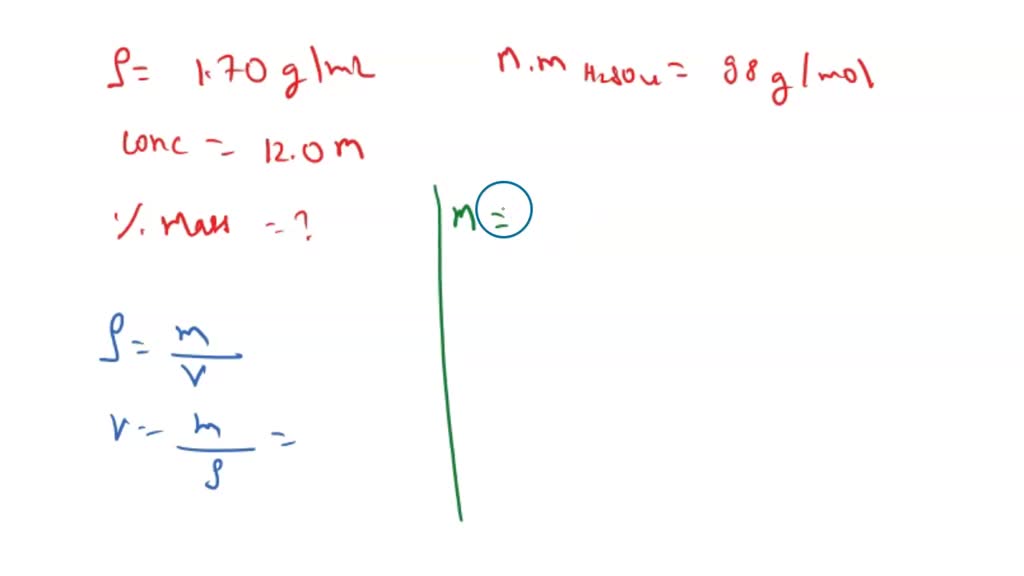
SOLVED: 1. A sample of Sulfuric acid is 12.0 M and has a density of 1.70 g/ml. Find mass % of sulfuric Acid (Molar mass of H2SO4 = 98 g/mol) 2. 250-gram

Calculate molecular mass of the following molecules: a. Sulphuric acid `(H_(2) SO_(4))` b. Gluco... - YouTube
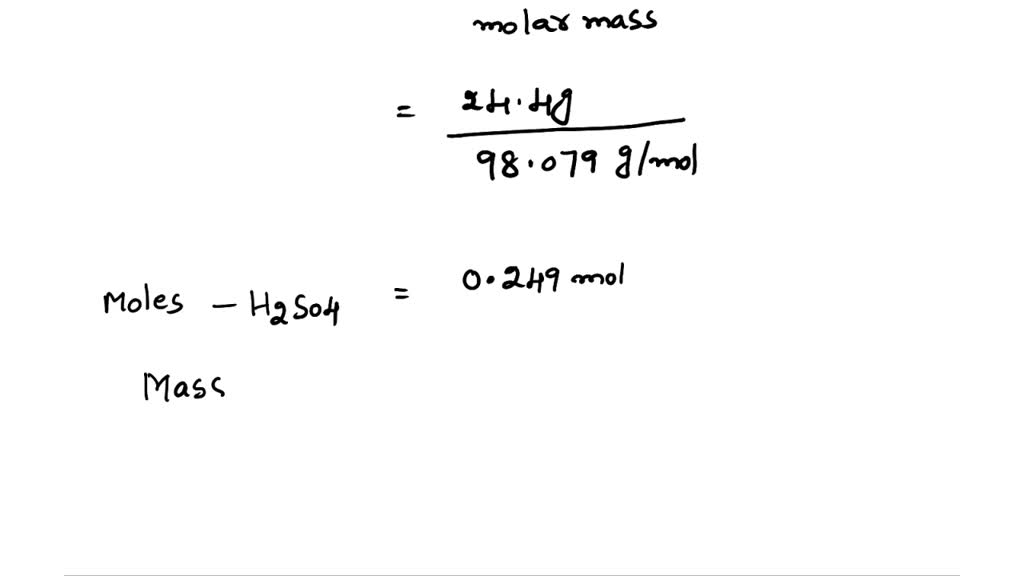
SOLVED: Calculate the molality of a sulfuric acid solution containing 24.4 g of sulfuric acid in 198 g of water. The molar mass of sulfuric acid is 98.09 g.

The molecular mass of H2SO4 is 98 amu. Calculate the number of moles of each elements in 294 g of H2SO4

Calculate the mass percent `(w//w)` of sulphuric acid in a solution prepared by dissovles 4 g of - YouTube

Question Video: Determining the Concentration of Sulfuric Acid Via Titration with Sodium Carbonate | Nagwa

molar mass of sulfuric acid/molecular mass/molecular weight of sulphuric acid/ H2SO4 molar mass. - YouTube
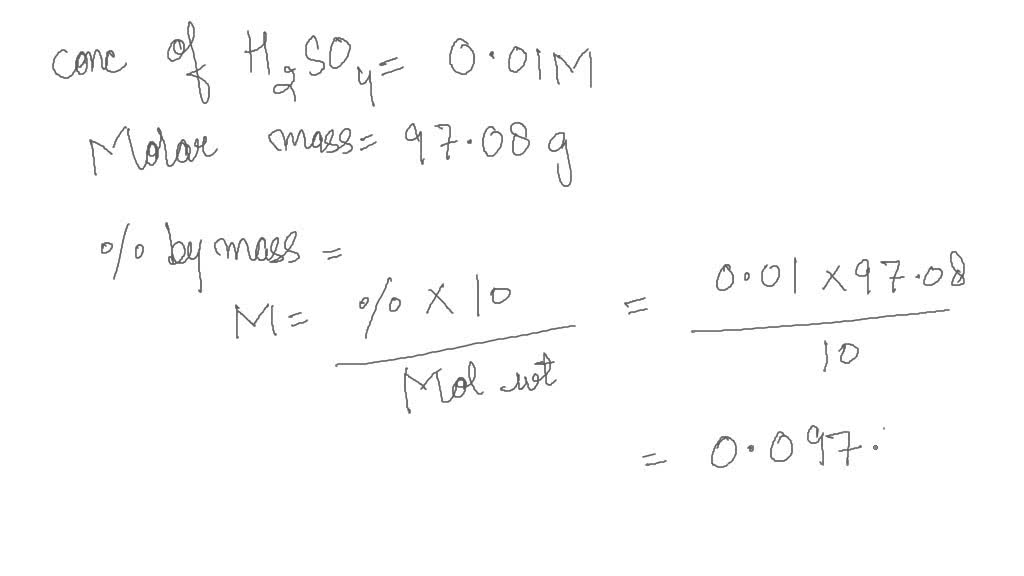
SOLVED: The molarity of a sulfuric acid solution is 0.01 M. Express this concentration in % by mass, ppm, and ppb.(Density of water is 1 g/mL; molar mass of sulfuric acid is
28. concentrated sulphuric acid has a density of 1.9 g/ml and is 99 percent h2so4 by weight calculate molarity of h2so4 in this acid