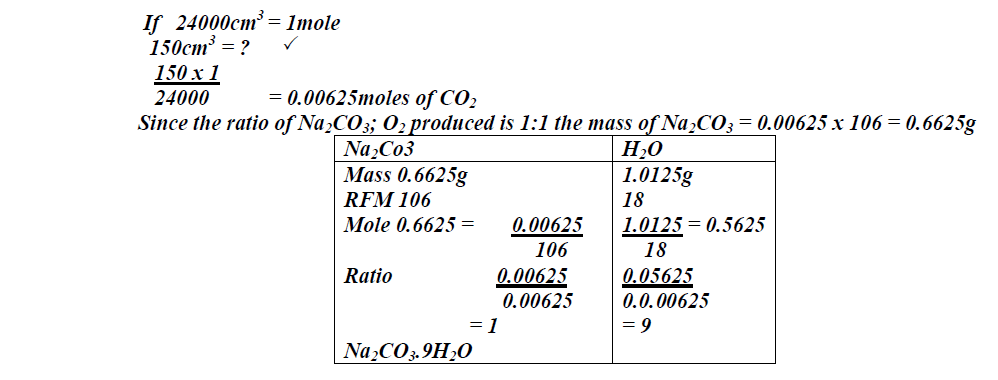
When 1.675g of hydrated sodium carbonate was reacted with excess hydrochloric acid, the volume carbon (IV) oxide gas obtained at room temperature and pressure was 150cm<sup>3</sup>). Calculate...

Sodium Carbonate + Hydrochloric Acid - Na2CO3 + HCl - Molecular Equations & Net Ionic Equations - YouTube

a). What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced - YouTube
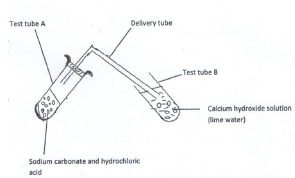
Study the diagram below for the reaction between sodium carbonate and hydrochloric acid and answer the questions that follow

35. Answer the following questions: (1) State the reaction of sodium carbonate and sodium hydrogen - Brainly.in

Sodium Carbonate + Hydrochloric Acid - Balanced Molecular and Net Ionic Equation - Na2CO3 + HCl | Quizalize
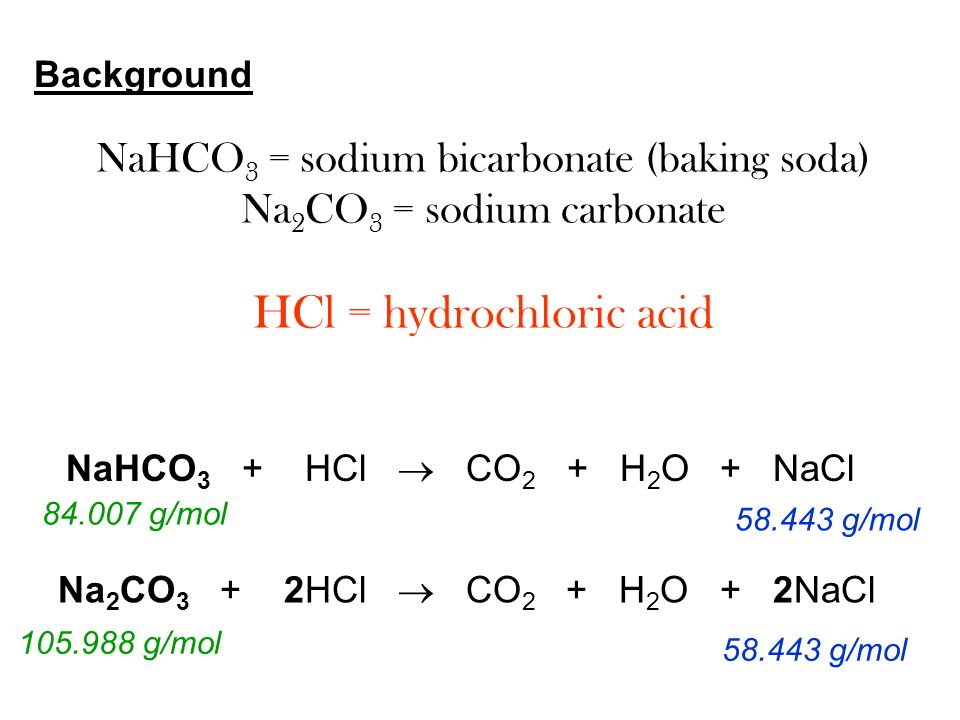
Lab 8 Sodium Carbonate or Sodium Bicarbonate? Objective To determine a compound to be either Na 2 CO 3 or NaHCO ppt download
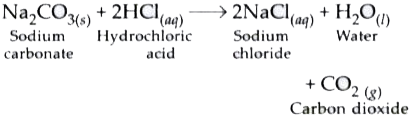
Write a balanced chemical equation for the reaction between sodium carbonate and hydrochloric acid indicating the physical state of the reactants and the products.
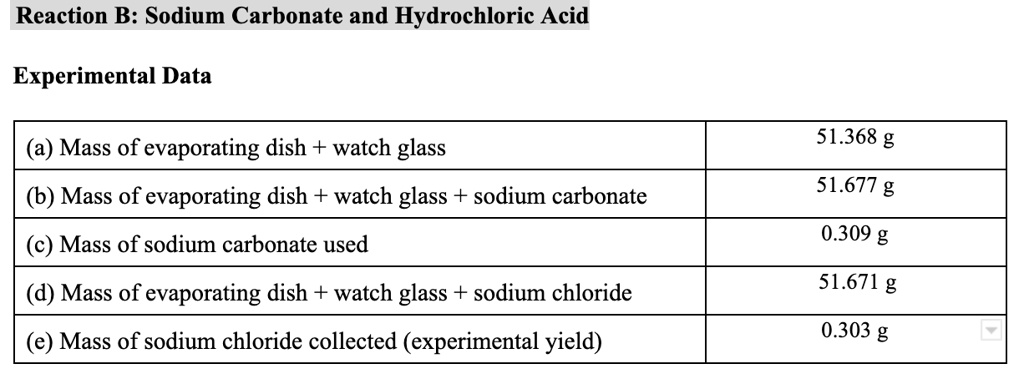
SOLVED: Reaction B: Sodium Carbonate and Hydrochloric Acid Experimental Data Mass of evaporating dish + watch glass 51.368 g Mass of evaporating dish + watch glass + sodium carbonate 51.677 g Mass

SOLVED: Reaction of Sodium carbonate (NazCOs) with hydrochloric acid Reaction NaCO and HCI Observalion for (he reacuon bobbles frm very fst Kazn Balance thc chemical equation LNA,CO, (4q) 2HCI (8q) 2NACl (#Q)

Question Video: Identifying the Observations of the Reaction between Hydrochloric Acid and Sodium Carbonate | Nagwa

![ANSWERED] Sodium carbonate and hydrochloric acid re... - Physical Chemistry ANSWERED] Sodium carbonate and hydrochloric acid re... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/51622204-1659180079.3710194.jpeg)