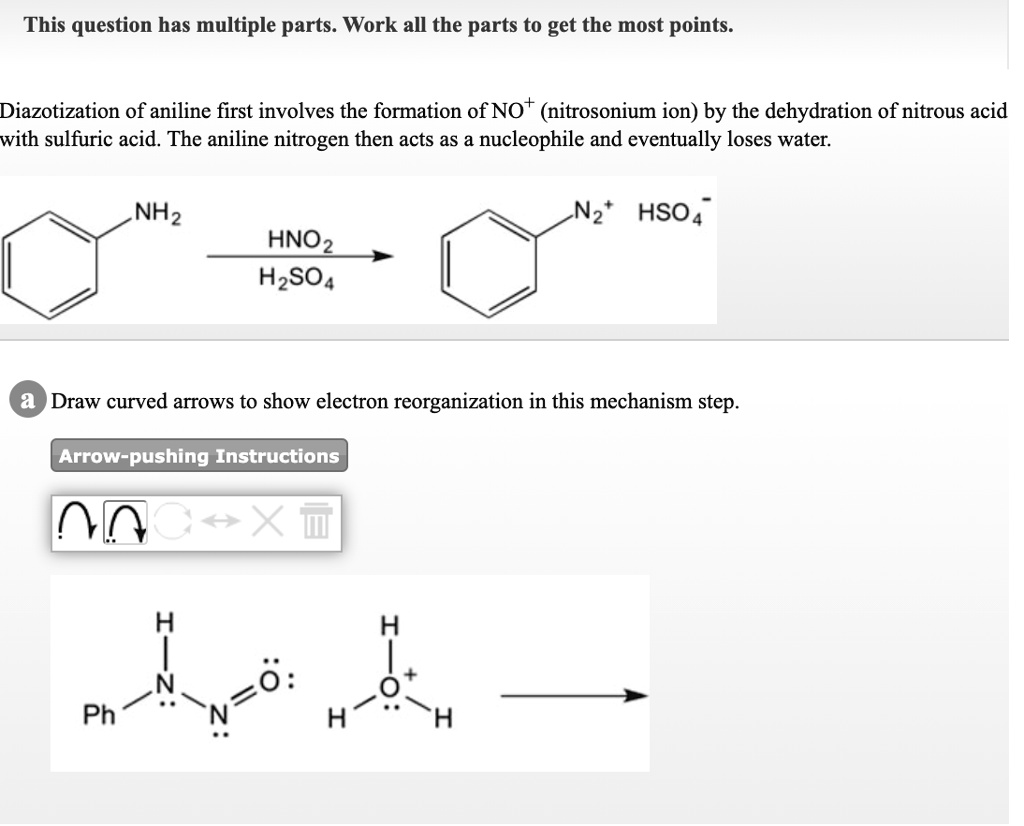
SOLVED: This question has multiple parts. Work all the parts to get the most points: Diazotization of aniline first involves the formation of NO+ (nitrosonium ion) by the dehydration of nitrous acid

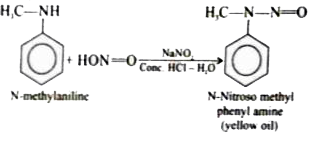
Give balanced equations for the following reactions:(i) Aniline is treated with nitrous acid and HCl at low temperature.(ii) Acetyl chloride is treated with ethyl alcohol.(iii) Formaldehyde is treated with ammonia.

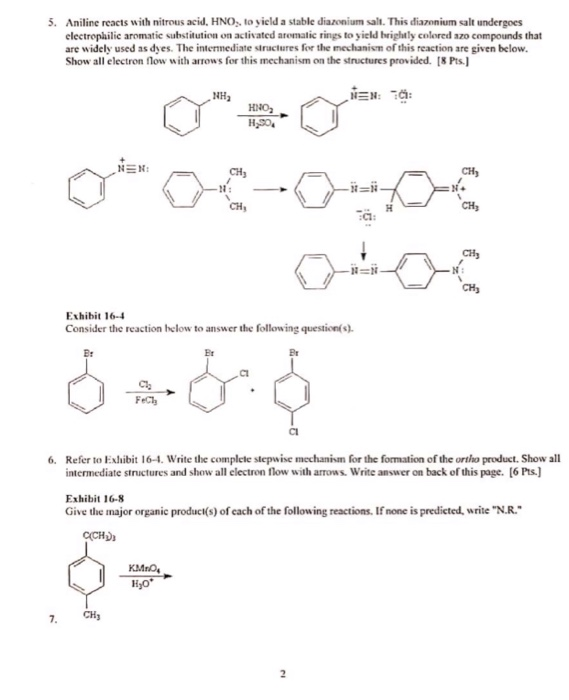
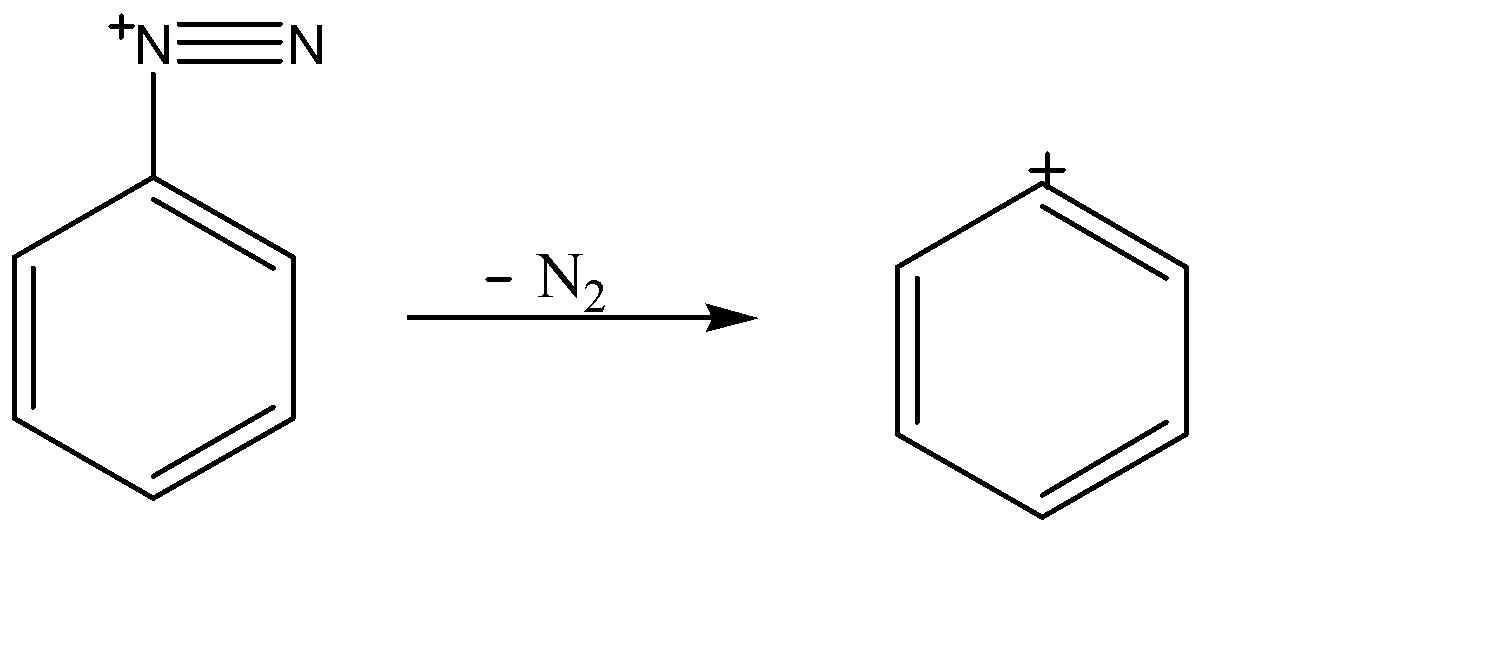
The diazotization of aniline first involves the formation of NO+ (nitrosonium ion) by the dehydration of nitrous acid with sulfuric acid. The aniline nitrogen then acts as a nucleophile and eventually loses
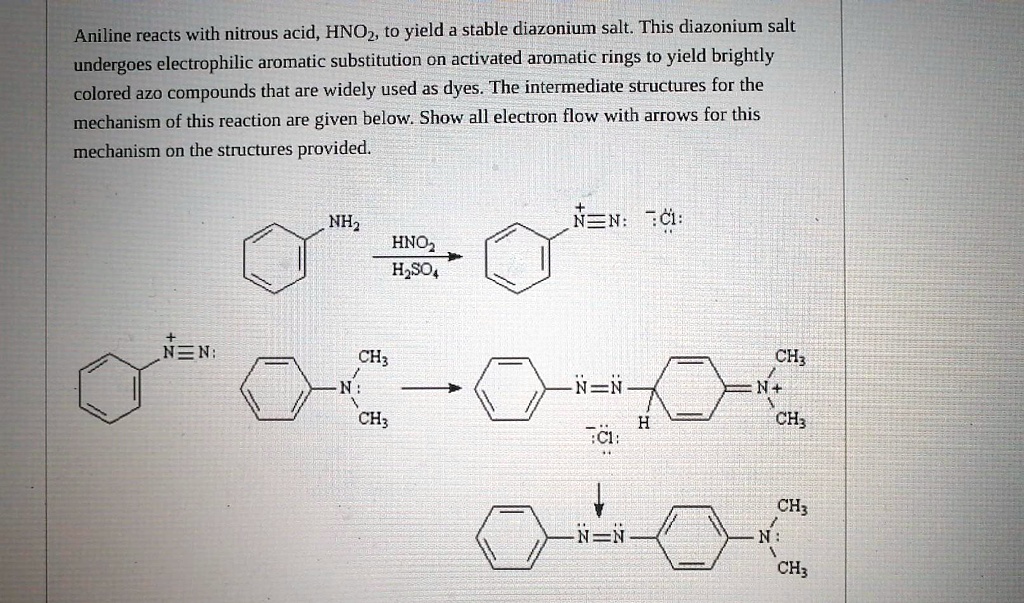
SOLVED: Aniline reacts with nitrous acid, HNOz to yield stable diazonium salt: This diazonium salt undergoes electrophilic aromatic substitution on activated aromatic rings t0 yield brightly colored az0 compounds that are widely
What is the action of nitrous acid on aniline? - Sarthaks eConnect | Largest Online Education Community
![Reaction of aniline with \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] followed by treatment of dilute acid gives: A) \\[{{\\text{C}}_{\\text{6}}}{{\\text{H}}_{\\text{5}}}{\\text{NHOH}}\\]B) \\[{{\\text{C}}_{\\text{6}}}{{\\text{H}}_{\\text{5}}}{\\text{OH ... Reaction of aniline with \\[{\\text{HN}}{{\\text{O}}_{\\text{2}}}\\] followed by treatment of dilute acid gives: A) \\[{{\\text{C}}_{\\text{6}}}{{\\text{H}}_{\\text{5}}}{\\text{NHOH}}\\]B) \\[{{\\text{C}}_{\\text{6}}}{{\\text{H}}_{\\text{5}}}{\\text{OH ...](https://www.vedantu.com/question-sets/a49c0b64-6198-4deb-b5a6-8fea6ba879417558707394620197326.png)



![Aniline reacts with conc. \\[\\;{\\text{HN}}{{\\text{O}}_{\\text{3}}}\\] to give:-\n \n \n \n \n Aniline reacts with conc. \\[\\;{\\text{HN}}{{\\text{O}}_{\\text{3}}}\\] to give:-\n \n \n \n \n](https://www.vedantu.com/question-sets/f102ae28-5724-46a5-be68-375d93b118677418549098762929594.png)


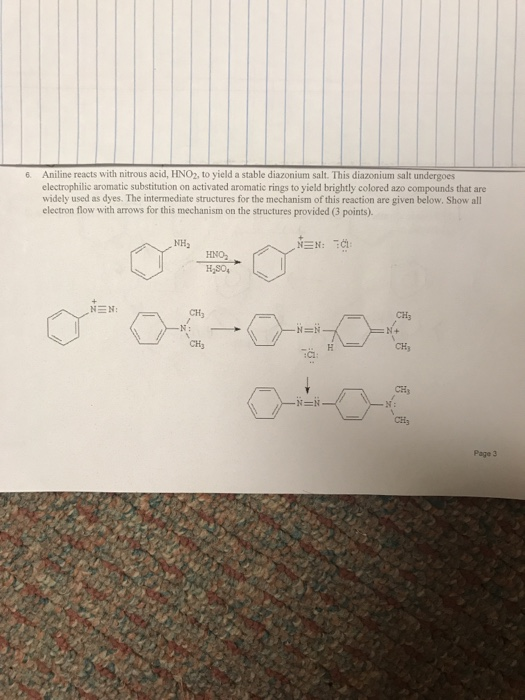
![Solved] Aniline Reacts with Nitrous Acid,HNO2,to Yield a Stable Diazonium Salt | Quiz+ Solved] Aniline Reacts with Nitrous Acid,HNO2,to Yield a Stable Diazonium Salt | Quiz+](https://d2lvgg3v3hfg70.cloudfront.net/TB4944/11eab917_1506_4469_99e6_1f99a6c80b57_TB4944_00.jpg)