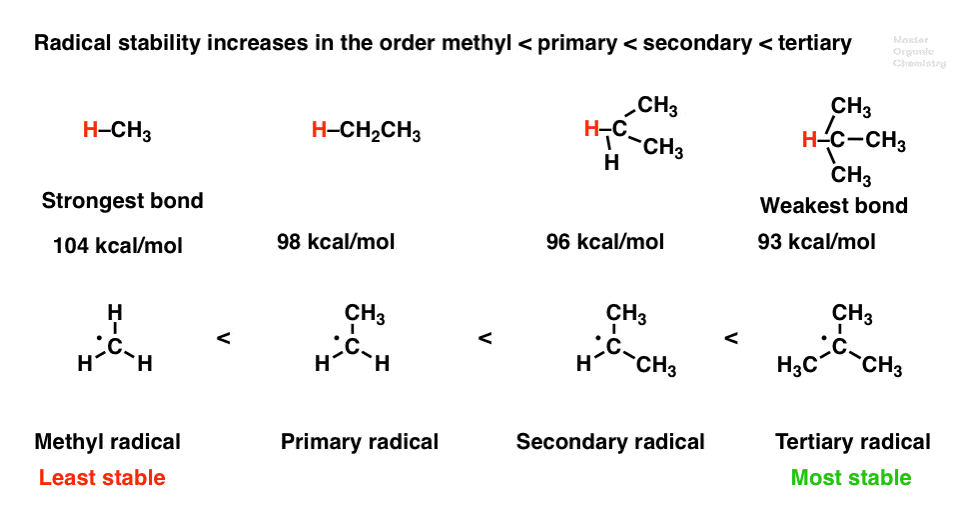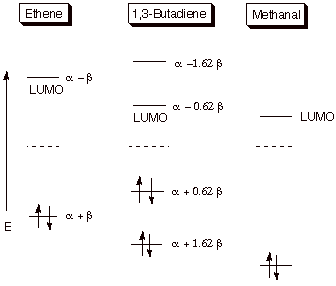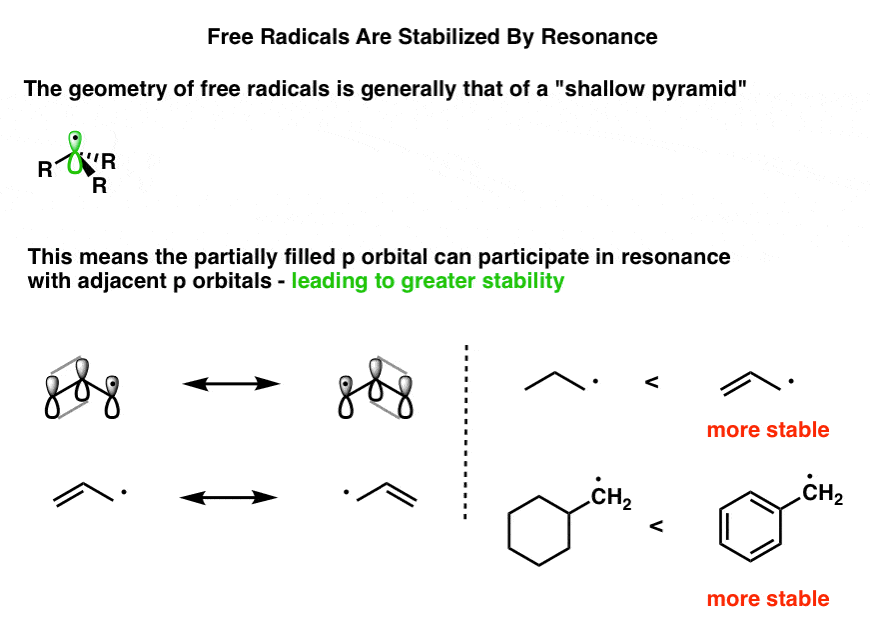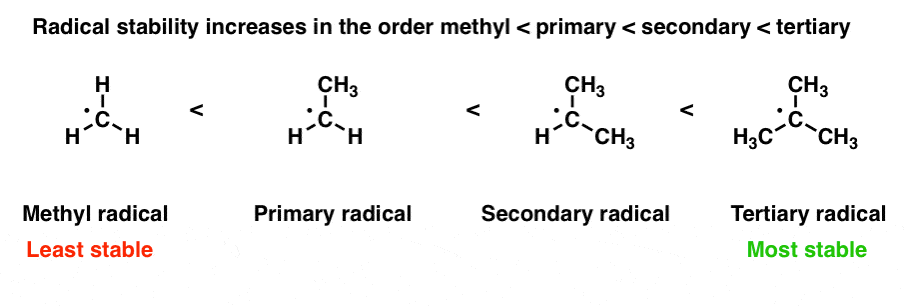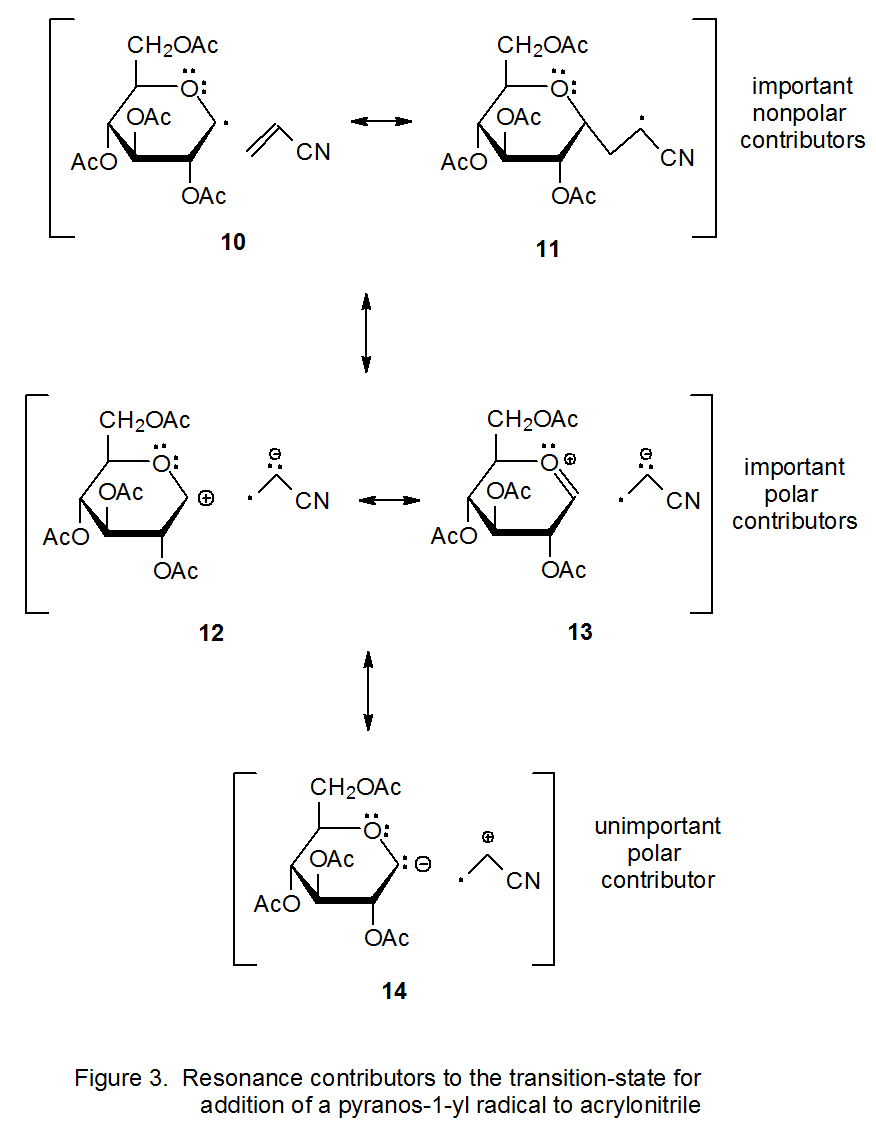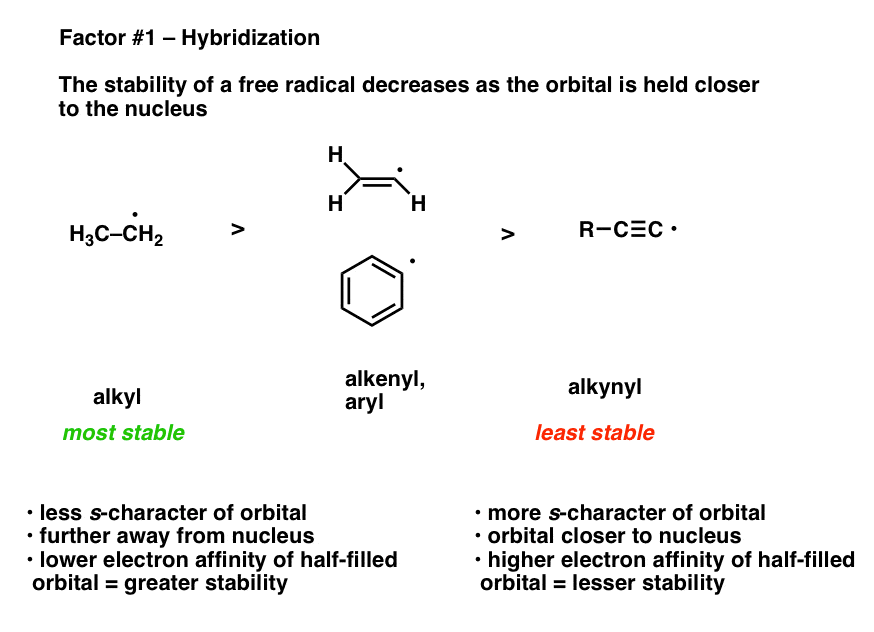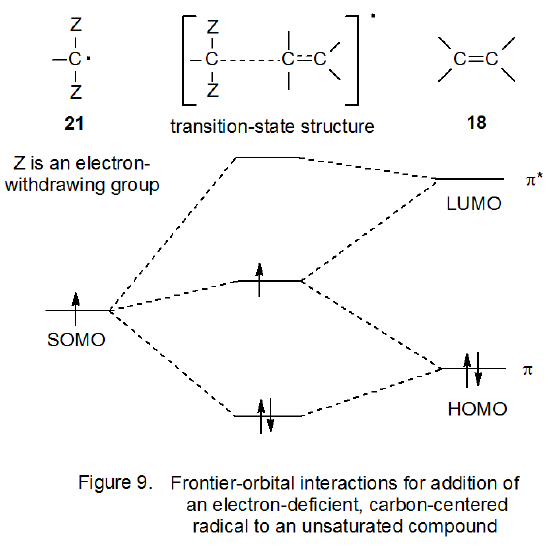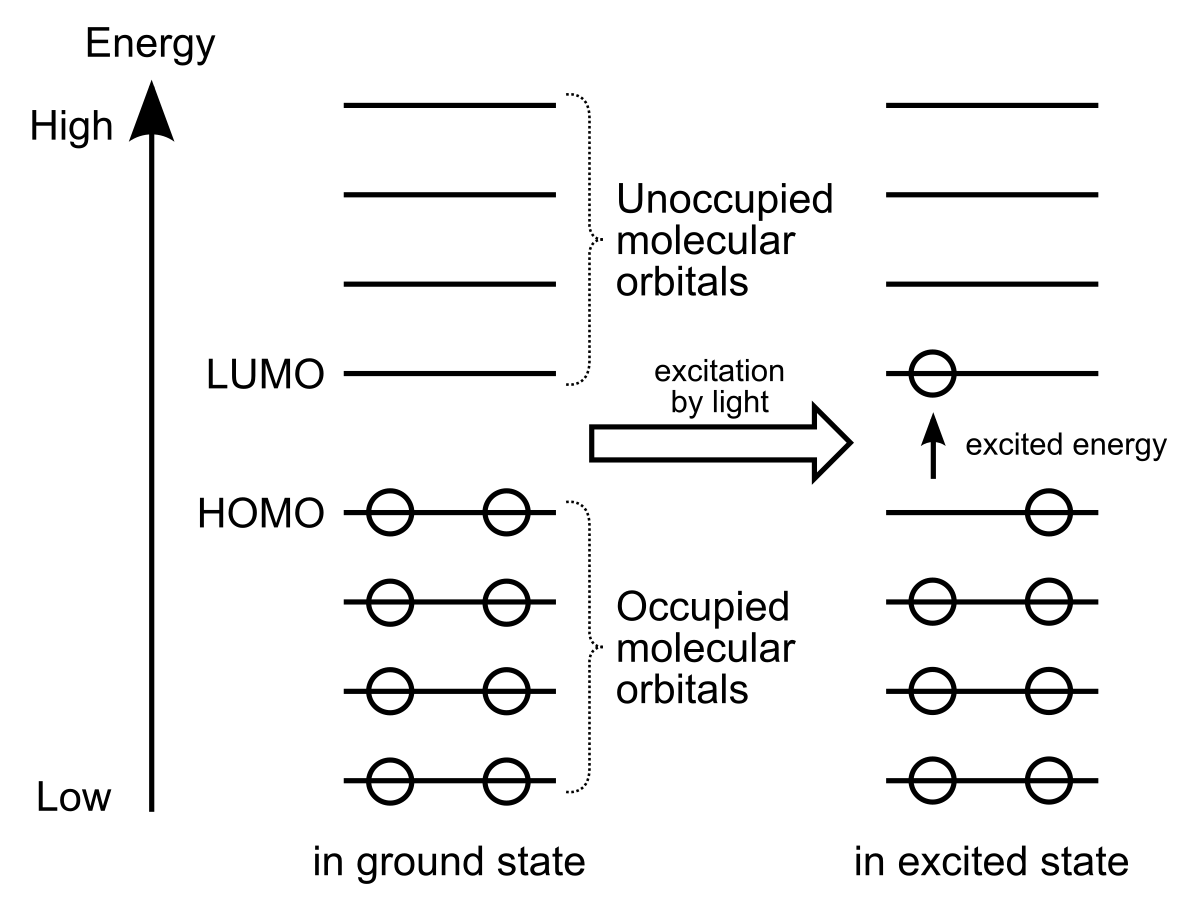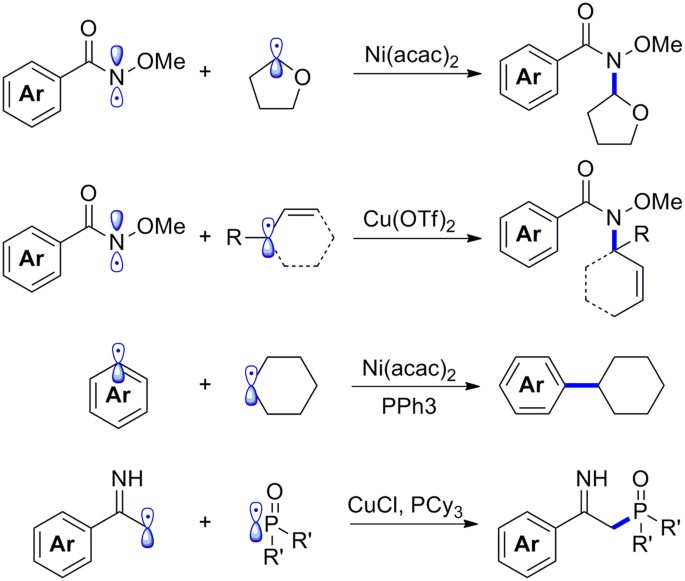
Stabilization of Two Radicals with One Metal: A Stepwise Coupling Model for Copper-Catalyzed Radical–Radical Cross-Coupling | Scientific Reports
HOMO and LUMO images of structural radicals 1-16 in all studied mediums... | Download Scientific Diagram
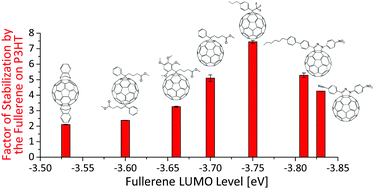
Influence of the fullerene LUMO level on the stability of bulk heterojunction solar cells - Journal of Materials Chemistry C (RSC Publishing)

Boron-centered stable radical anion/radical cation pair. Reproduced... | Download Scientific Diagram

Theoretical basis for the stabilization of charges by radicals on electrified polymers - Chemical Science (RSC Publishing) DOI:10.1039/C6SC02672A

Molecules | Free Full-Text | Synthesis, Physical Properties, and Reactivity of Stable, π-Conjugated, Carbon-Centered Radicals

A Simple Approach to Achieve Organic Radicals with Unusual Solid-State Emission and Persistent Stability | CCS Chemistry
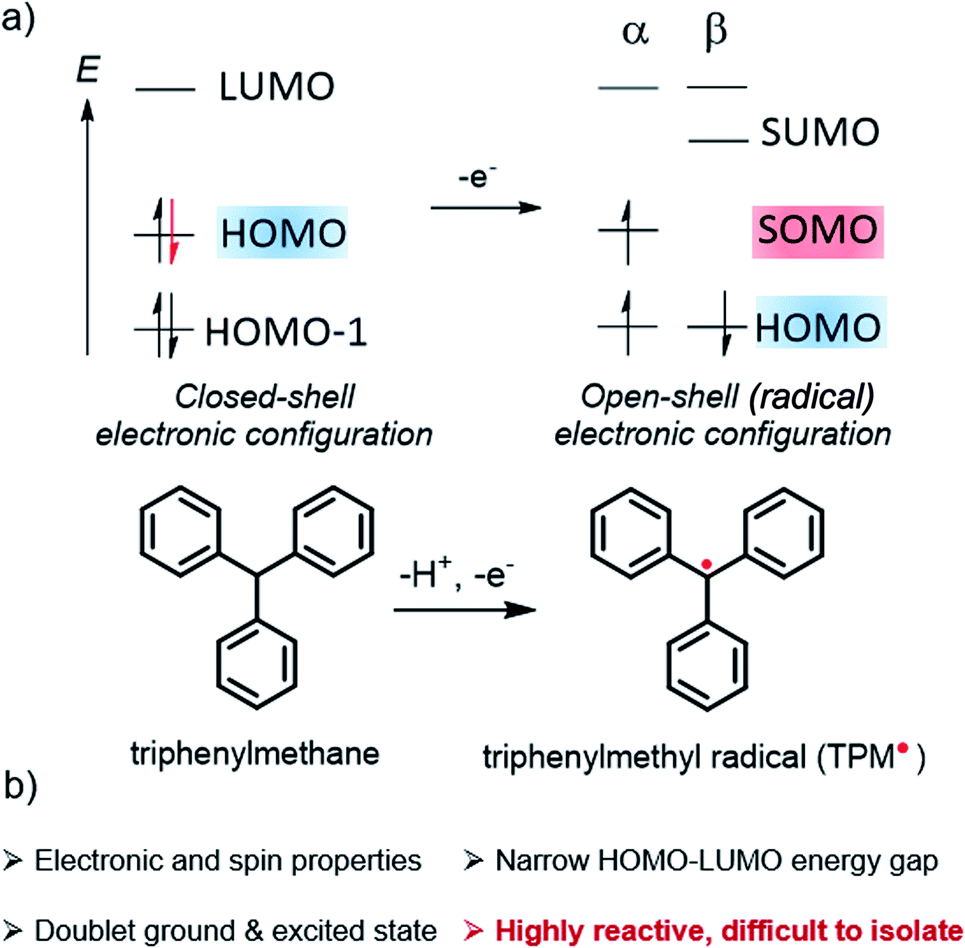
Organic radicals with inversion of SOMO and HOMO energies and potential applications in optoelectronics - Chemical Science (RSC Publishing) DOI:10.1039/D2SC02480B
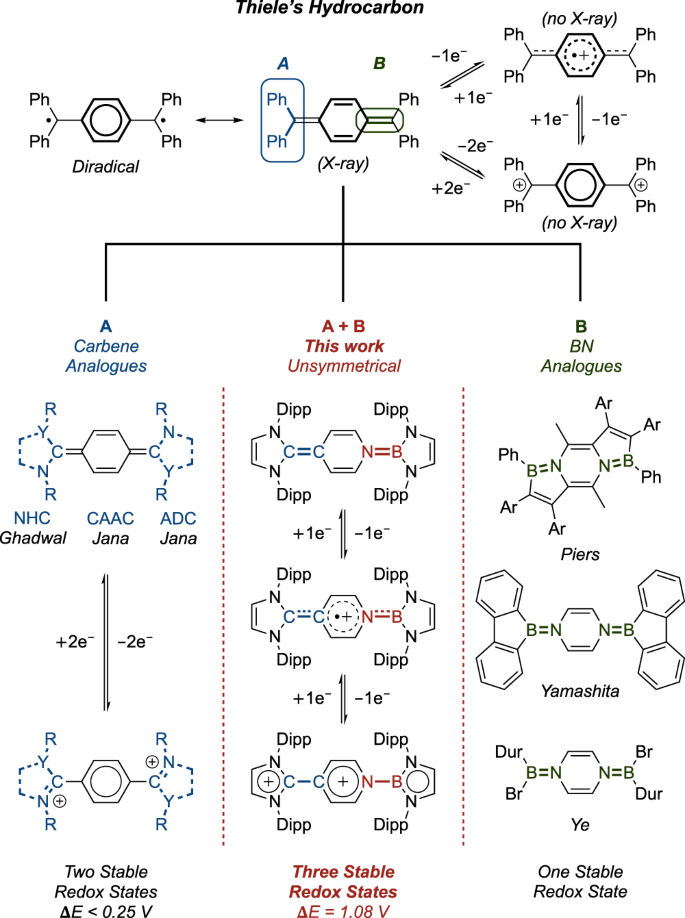
A crystalline radical cation derived from Thiele's hydrocarbon with redox range beyond 1 V | Nature Communications
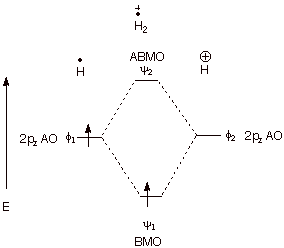
1. Strong Covalent Bonds. Consider the pi bond of ethene in simple molecular orbital terms (The qualitative results would be the same for any pi or sigma bond.

Depictions of the HOMO (red/blue) and LUMO (green/ yellow) of P, 1 + ,... | Download Scientific Diagram

Efficient light-emitting diodes from organic radicals with doublet emission: Journal of Applied Physics: Vol 129, No 18

A supramolecular strategy for tuning the energy level of naphthalenediimide: Promoted formation of radical anions with extraordinary stability - Chemical Science (RSC Publishing) DOI:10.1039/C5SC00862J

Highly Stable 1,2-Dicarbonyl Radical Cations Derived from N-Heterocyclic Carbenes | Journal of the American Chemical Society

SOLVED: CHAPTER Five Organic Chemistry EMiStRY Orbitale Combinations Table 5.3 Possible Frontier Molecular ( Examples of' Typical Qutcome HOMO LUMO ofS ! substitution Bond = formation in step or free radical addition

Fluorescent Organic π‐Radicals Stabilized with Boron: Featuring a SOMO–LUMO Electronic Transition - Ito - 2022 - Angewandte Chemie International Edition - Wiley Online Library