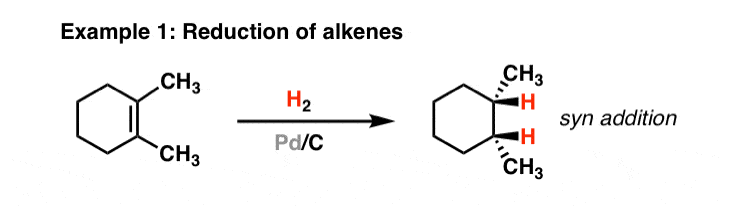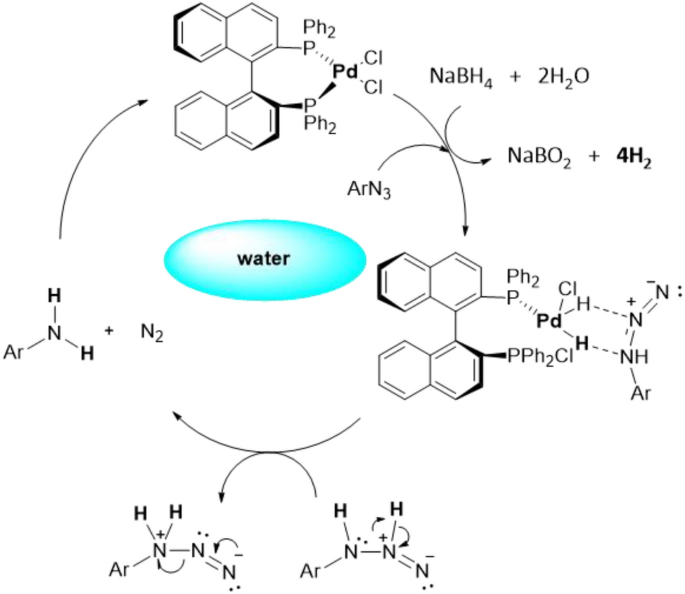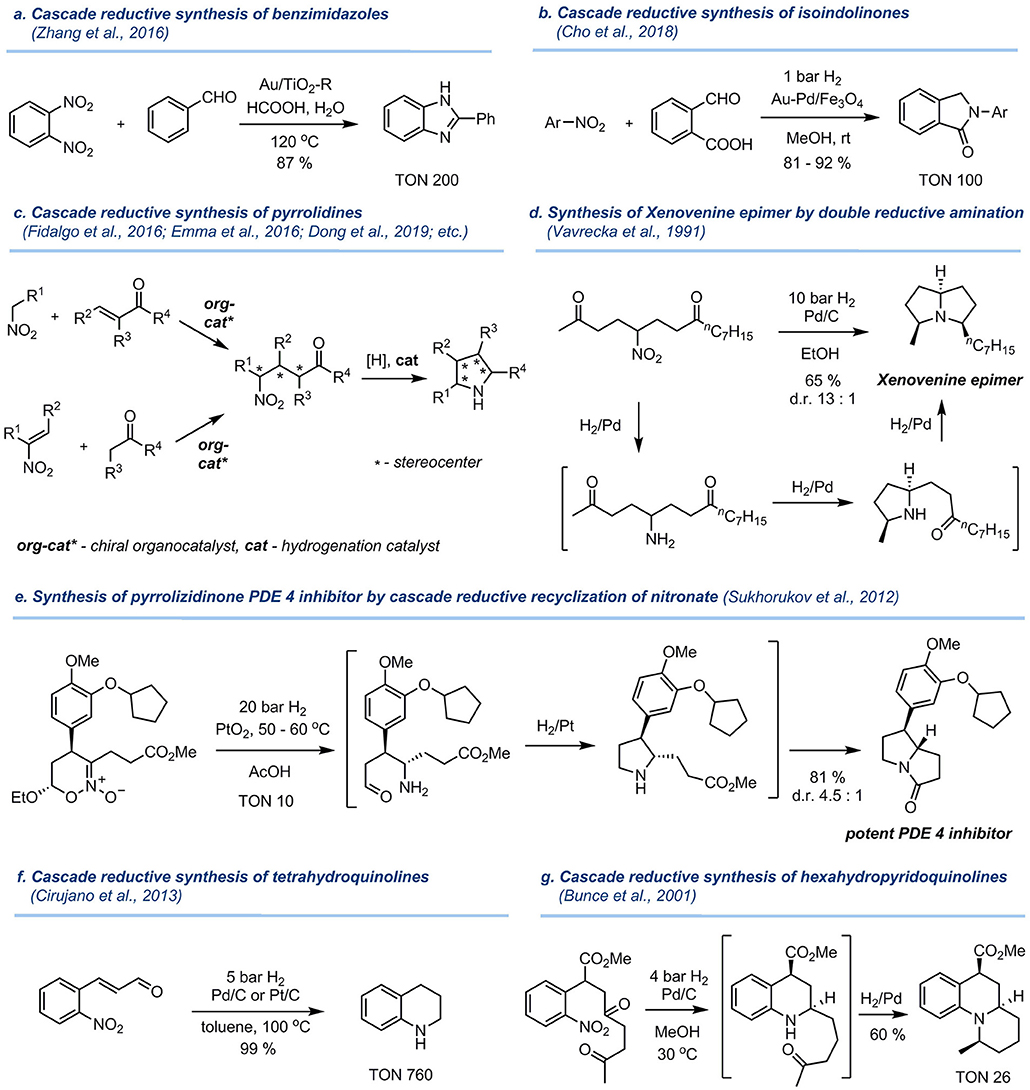
Frontiers | Catalytic Reductive Amination of Aldehydes and Ketones With Nitro Compounds: New Light on an Old Reaction

Full article: Highly efficient reduction of nitro compounds: Recyclable Pd/C-catalyzed transfer hydrogenation with ammonium formate or hydrazine hydrate as hydrogen source

Saline Accelerates Oxime Reaction with Aldehyde and Keto Substrates at Physiological pH | Scientific Reports

Commercial Pd/C-Catalyzed N-Methylation of Nitroarenes and Amines Using Methanol as Both C1 and H2 Source | The Journal of Organic Chemistry
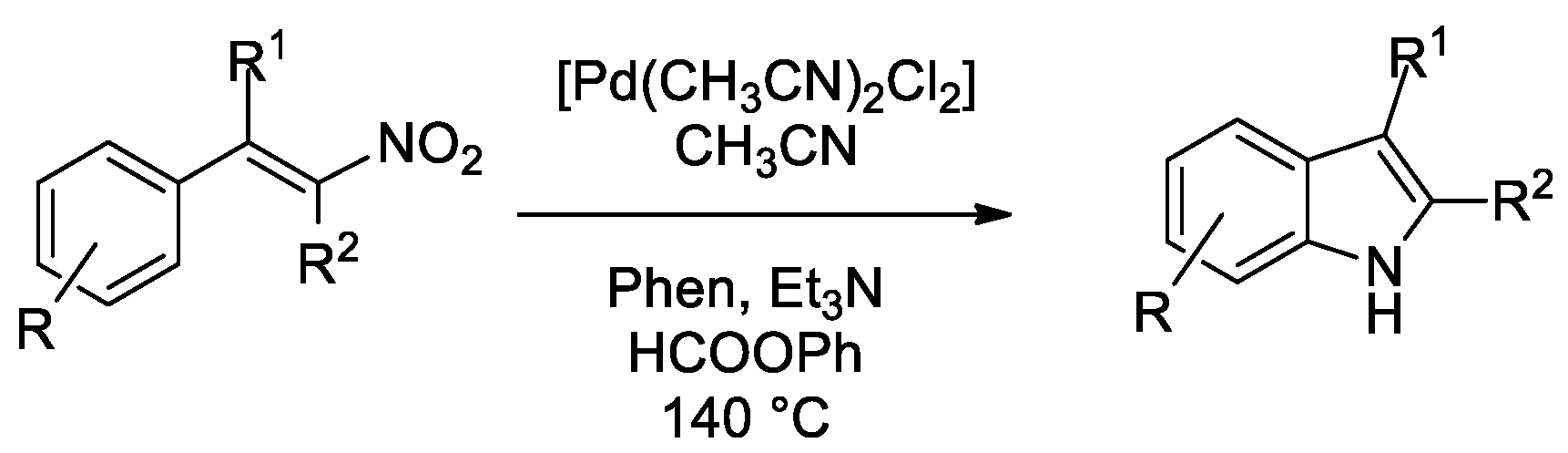
Catalysts | Free Full-Text | Phenyl Formate as a CO Surrogate for the Reductive Cyclization of Organic Nitro Compounds to Yield Different N-Heterocycles: No Need for Autoclaves and Pressurized Carbon Monoxide

Directed C-H Bond Oxidation of Bridged Cycloalkanes Catalyzed by Palladium(II) Acetate. | Semantic Scholar

Commercial Pd/C-Catalyzed N-Methylation of Nitroarenes and Amines Using Methanol as Both C1 and H2 Source | The Journal of Organic Chemistry

Transfer hydrogenation of nitroarenes using cellulose filter paper-supported Pd/C by filtration as well as sealed methods - RSC Advances (RSC Publishing) DOI:10.1039/D2RA01151D

PDF) A mild procedure for the production of secondary amines from oximes and benzisoxazoles | Petros G Tsoungas - Academia.edu

Pd(0)-Catalyzed Intramolecular Reductive Heck Reaction of Vinyl Iodide and Oxime Ether: Enantioselective Synthesis of Cyclic Allylic N-Alkoxy Amine | Organic Letters

Selective Synthesis of Primary Anilines from Cyclohexanone Oximes by the Concerted Catalysis of a Mg–Al Layered Double Hydroxide Supported Pd Catalyst | Journal of the American Chemical Society

Commercial Pd/C-Catalyzed N-Methylation of Nitroarenes and Amines Using Methanol as Both C1 and H2 Source | The Journal of Organic Chemistry
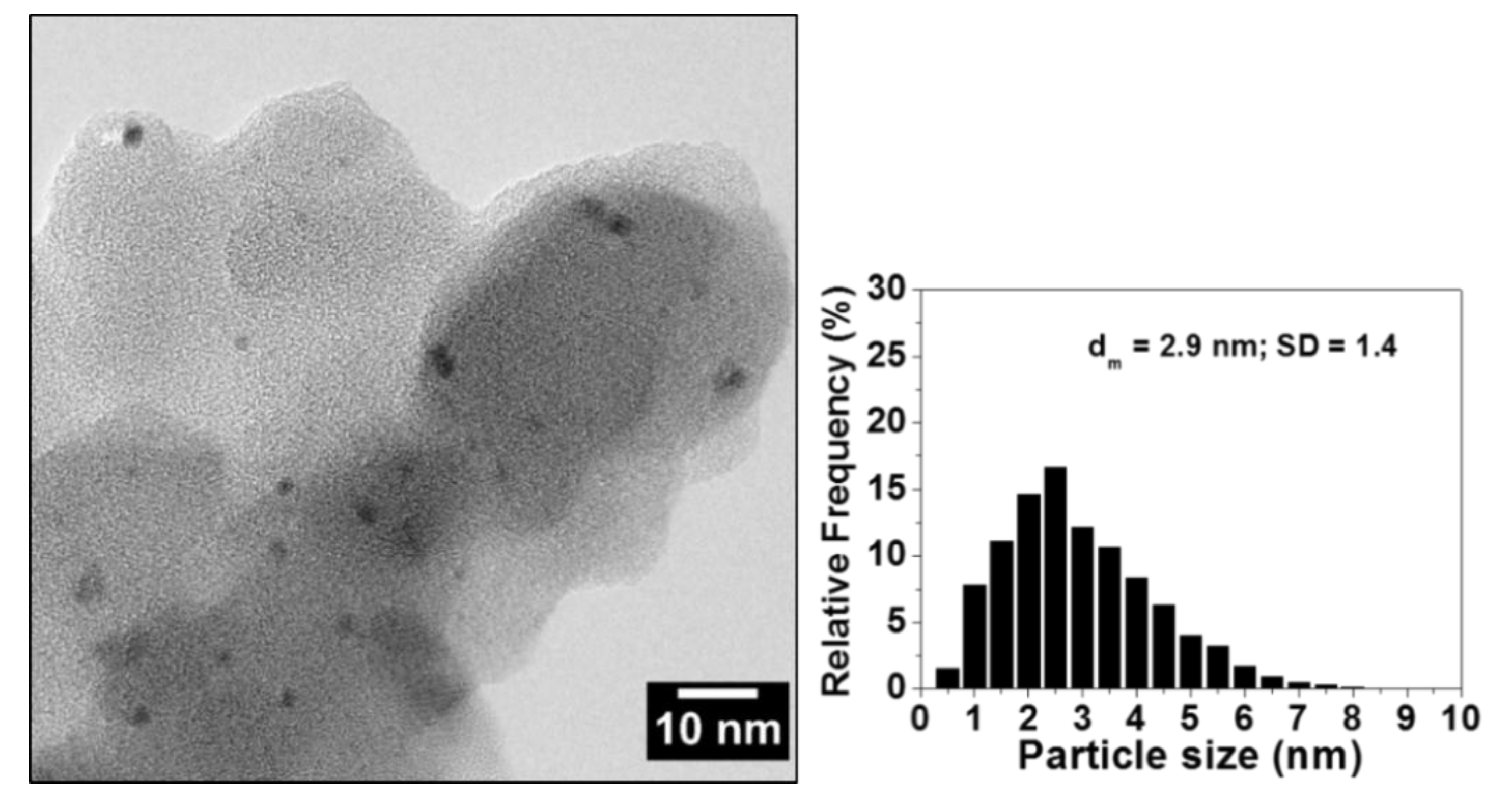
Catalysts | Free Full-Text | Polyvinylpyridine-Supported Palladium Nanoparticles: An Efficient Catalyst for Suzuki–Miyaura Coupling Reactions

PDF) ChemInform Abstract: Aryl-Pd Covalently Bonded Palladacycles, Novel Amino and Oxime Catalysts {Di-μ-chlorobis(benzaldehydeoxime-6-C,N)dipalladium(II), Di-μ-chlorobis(dimethylbenzylamine-6-C,N)dipalladium(II)} for the Heck reaction.

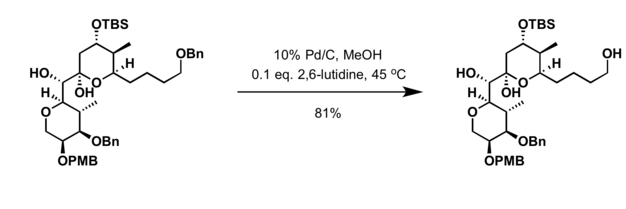

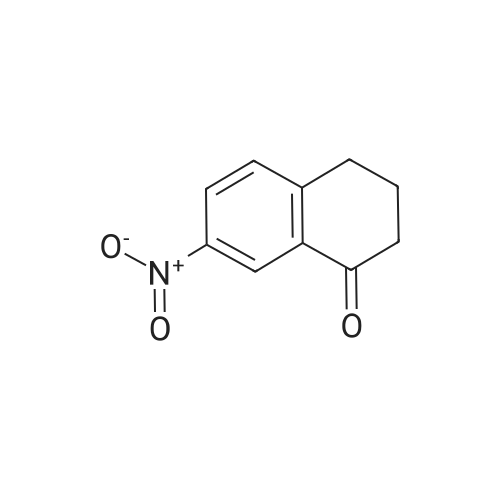
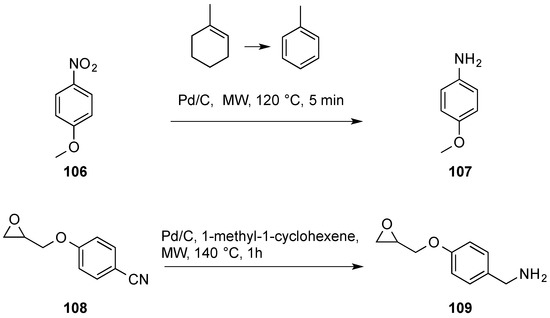
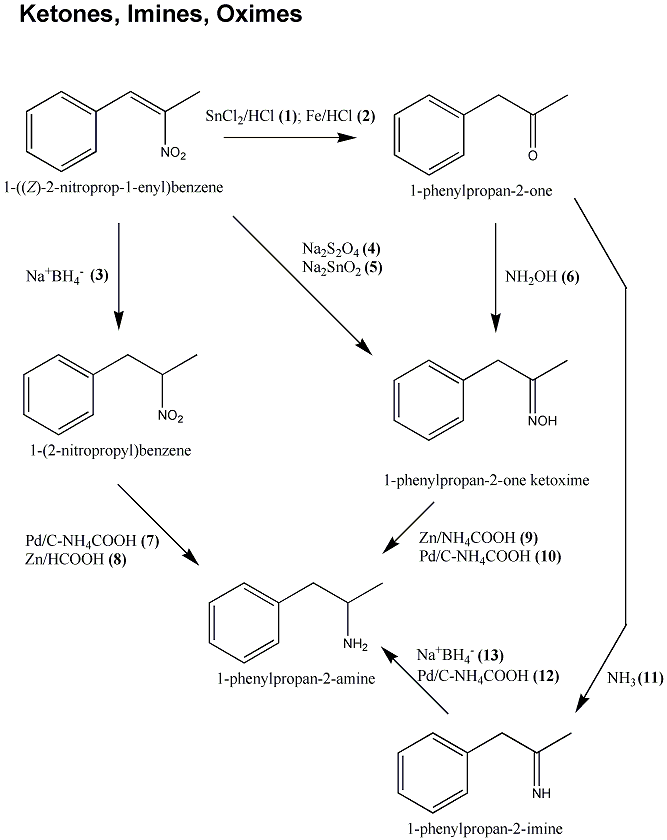
![Review: Catalytic Hydrogen Transfer Reductions Using Ammonium Formate - [www.rhodium.ws] Review: Catalytic Hydrogen Transfer Reductions Using Ammonium Formate - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/pictures/cth-af-fig2.gif)
![Oximes from Conjugated Nitroalkenes using Pd/C-Ammonium Formate CTH - [www.rhodium.ws] Oximes from Conjugated Nitroalkenes using Pd/C-Ammonium Formate CTH - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/pictures/ns2oxime-cth-tbl800.gif)