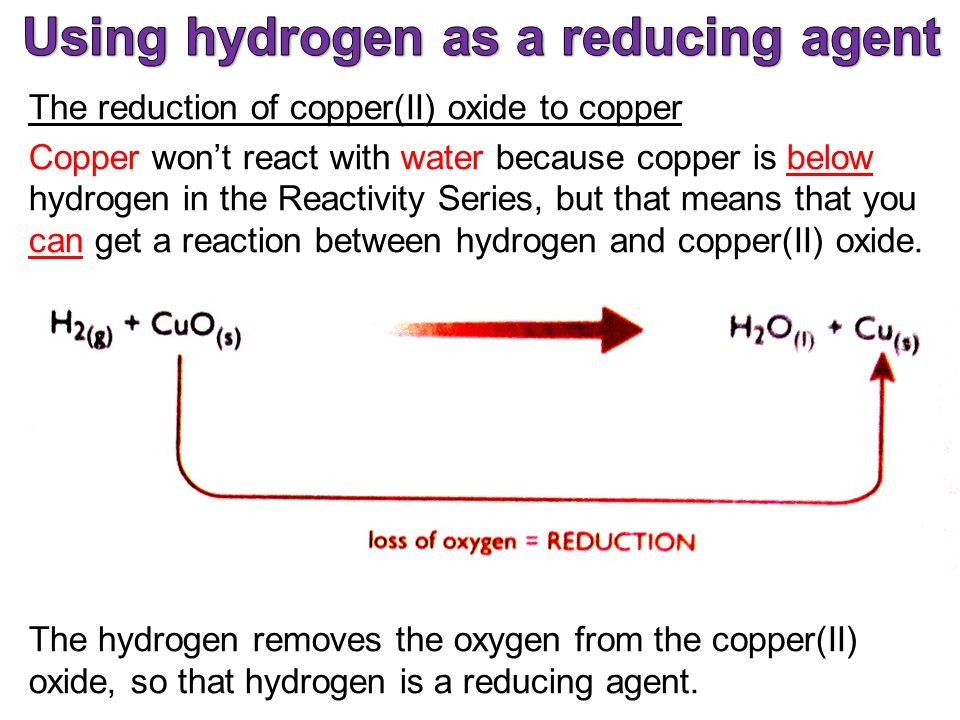
The set-up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide - Tutorke
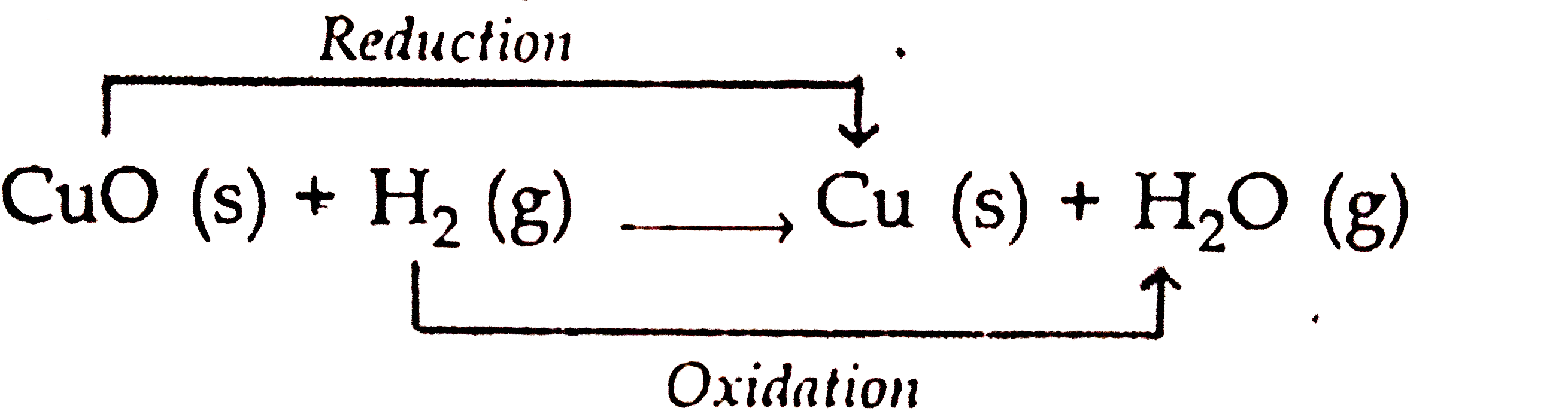
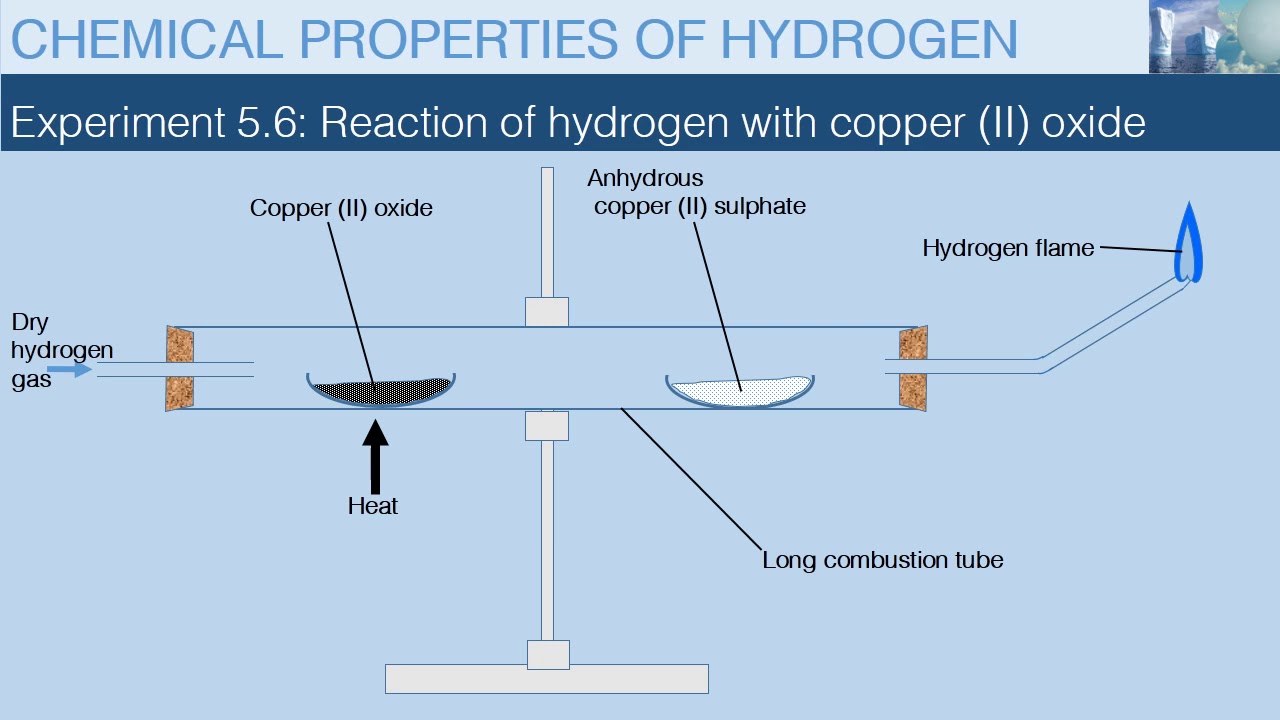
When hydrogen gas is passed over heated copper (II) oxide, copper and steam are formed. Write the balanced chemical equation for this reaction and state (i) the substance oxidized and (ii) the

Effects of Morphology and Surface Properties of Copper Oxide on the Removal of Hydrogen Sulfide from Gaseous Streams | Industrial & Engineering Chemistry Research

When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are : {:((i)"elements",(ii),"compounds",(iii),"reactants"),((iv)"products",(v),"metals",(vi),"non ...
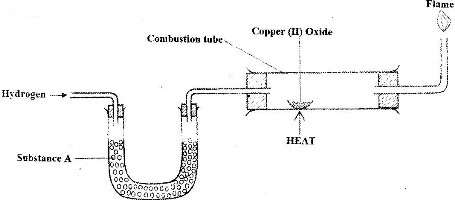
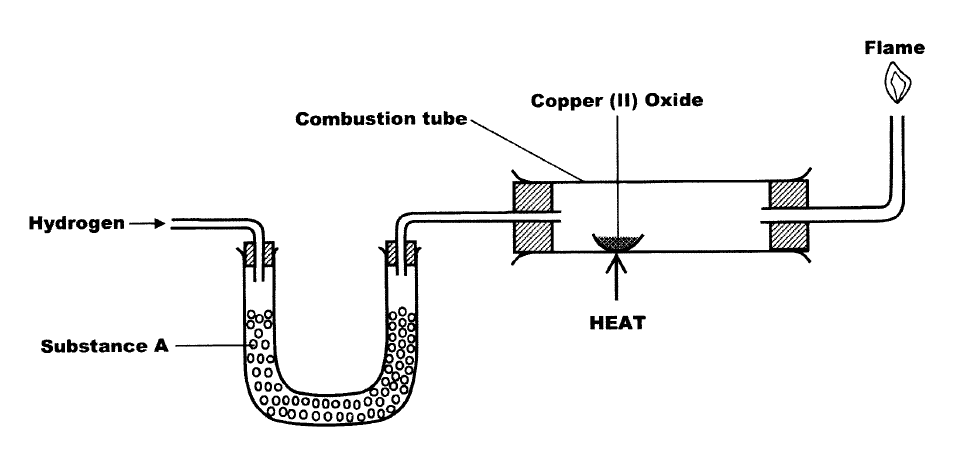
The set up below was used to investigate the reaction between dry hydrogen gas and copper (II) oxide.
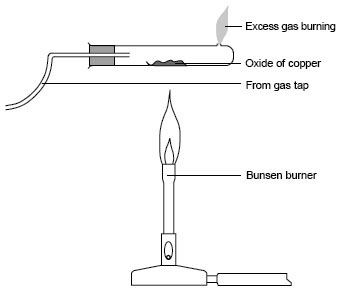
When copper (II) oxide is heated in a hard glass tube, and dry hydrogen is passed over, what colour change do you observe? - Quora

Treating Copper(II) Oxide Nanoflowers with Hydrogen Peroxide: A Novel and Facile Strategy To Prepare High‐Performance Copper(II) Oxide Nanosheets with Exposed (1 1 0) Facets - Peng - 2016 - ChemCatChem - Wiley Online Library
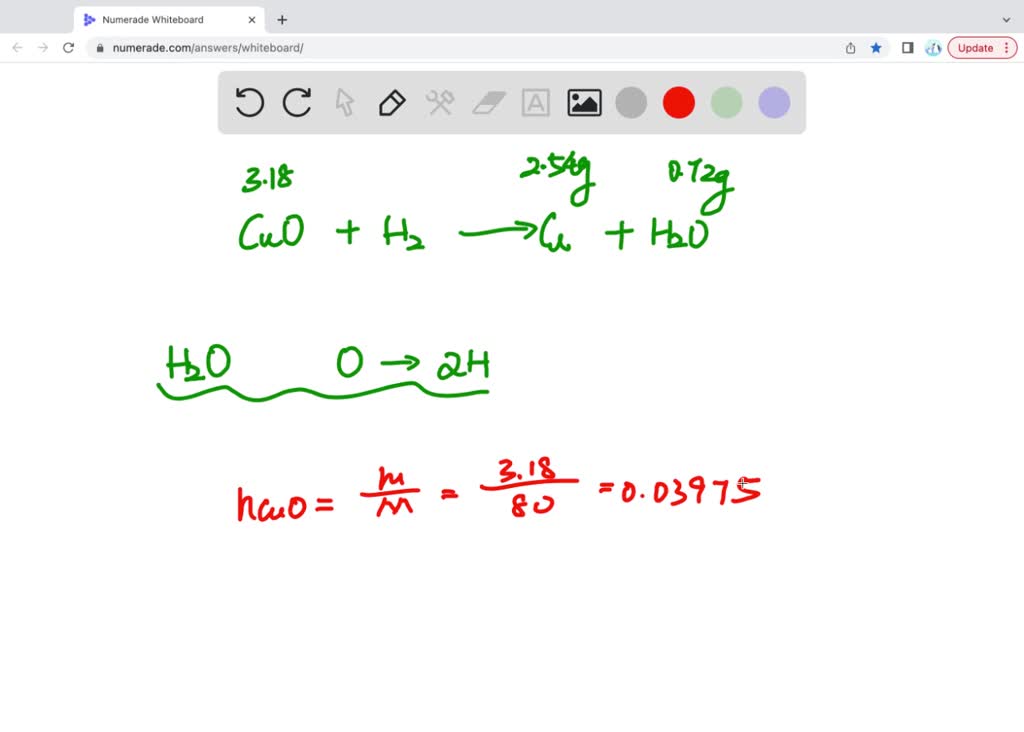
SOLVED: when 3.18 g of copper (||) oxide were carefully heated in a stream of dry hydrogen, 2.54 g of copper and 0.72 g of water were formed determine the number of