
PDF) Aqueous-Mediated Ring Opening of Epoxides with Oximes: A Rapid Entry into β-Hydroxy Oxime O-Ethers as Potential β-Adrenergic Blocking Agents

Mechanism of the Reaction of Olefins with Nitrous Anhydride (O═N–O–N═O) to Form 1,2-Oxazetes | Organic Letters

Molecules | Free Full-Text | Chemical Composition and in-Vitro Evaluation of the Antimicrobial and Antioxidant Activities of Essential Oils Extracted from Seven Eucalyptus Species
REACTIONS INDUCED BY CATION RADICALS AND STUDIES OF OXIDATION OF 0-NITROBENZENESELENENYL COMPOUNDS by SHISHUE CHIOU, B.S., M.S.

BINOL-Al catalysed asymmetric cyclization and amplification: preparation of optically active menthol analogs - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB00433K

Table 1 from Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis † | Semantic Scholar
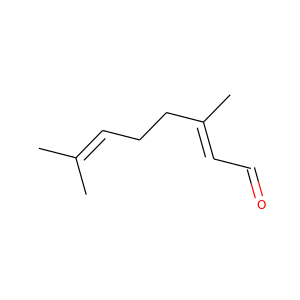
Thiofanocarb (75013-98-8, 39196-18-4) - Chemical Safety, Models, Suppliers, Regulation, and Patents - Chemchart

Oximes of 3,7-dimethylocta-2,6-dienal: Green synthesis, preparative separation of all diastereomers and complete assignment of 1H and 13C NMR spectra - ScienceDirect

Allergic Contact Dermatitis––Formation, Structural Requirements, and Reactivity of Skin Sensitizers | Chemical Research in Toxicology
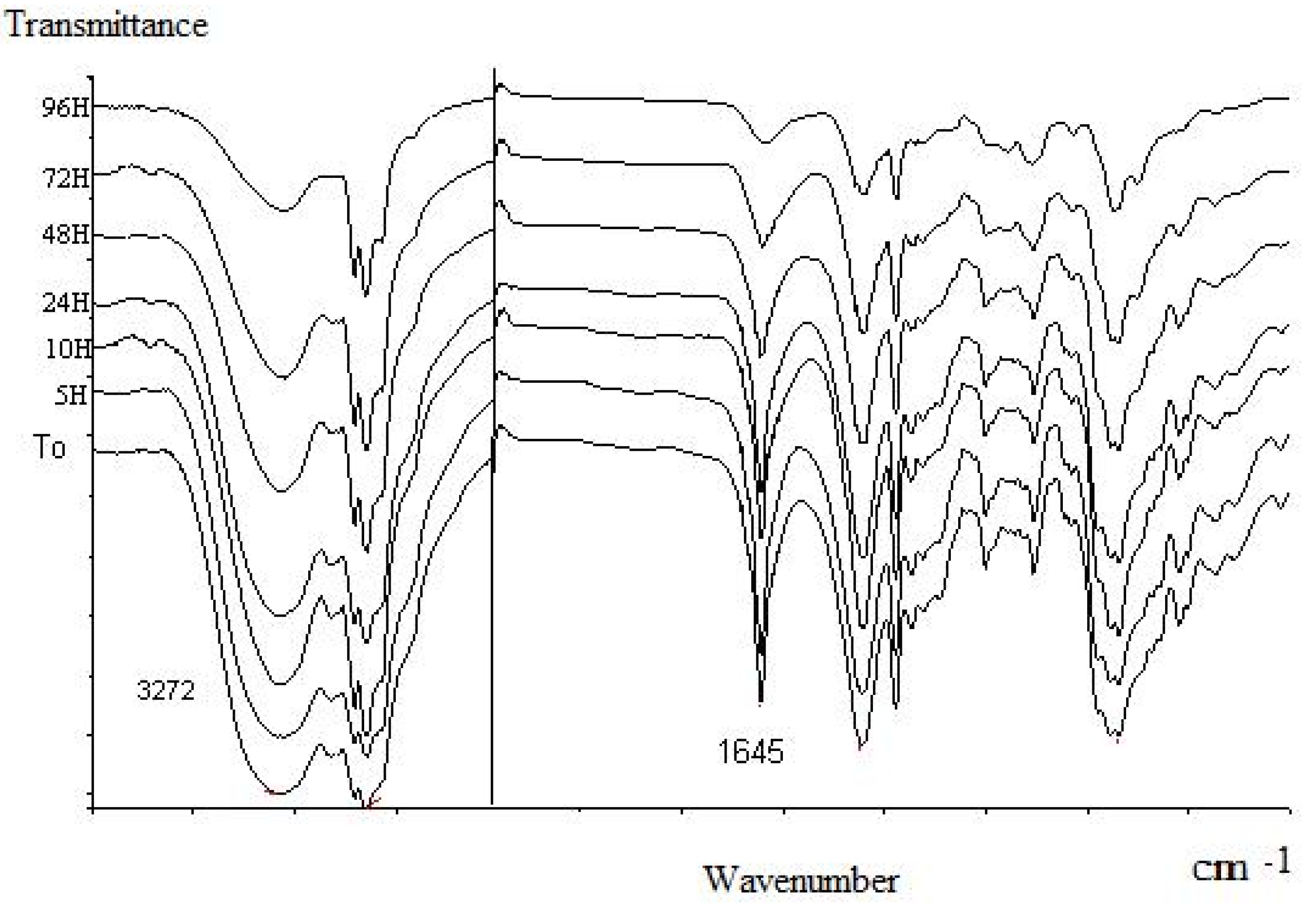
Molecules | Free Full-Text | Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †
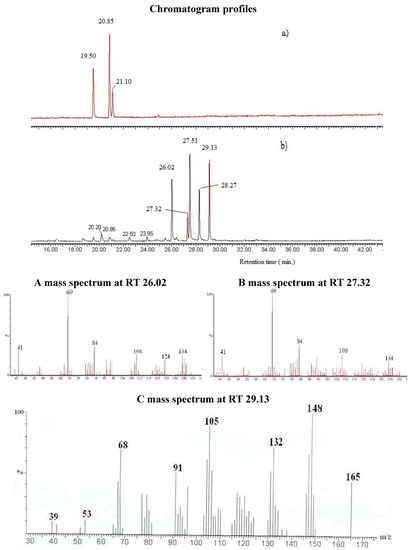
Molecules | Free Full-Text | Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †

Mass spectrum of Oxime- methoxy-phenyl with retention time (RT) = 3.504. | Download Scientific Diagram

Mass spectrum of Oxime- methoxy-phenyl with retention time (RT) = 3.504. | Download Scientific Diagram

PDF) Aqueous-Mediated Ring Opening of Epoxides with Oximes: A Rapid Entry into β-Hydroxy Oxime O-Ethers as Potential β-Adrenergic Blocking Agents









