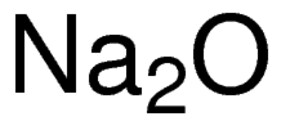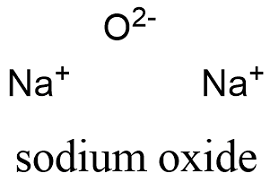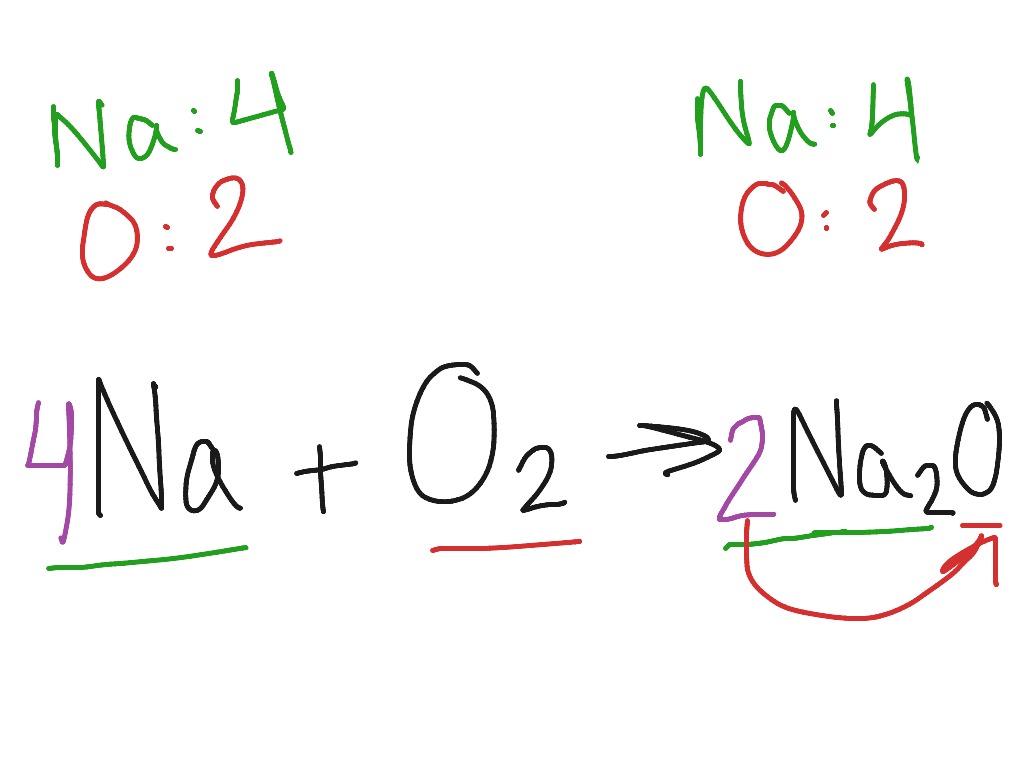inorganic chemistry - Is sodium superoxide more abundant than sodium oxide? - Chemistry Stack Exchange
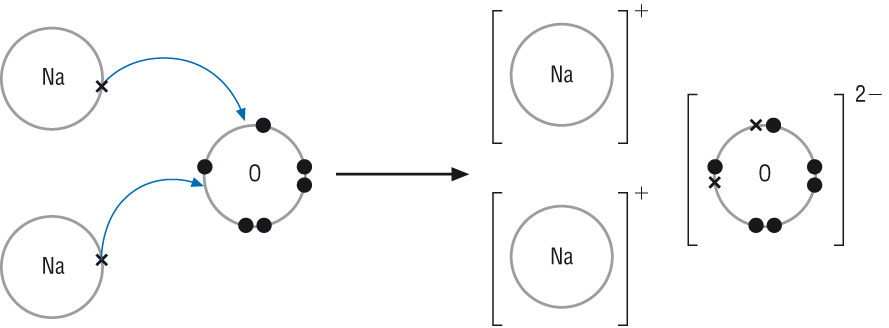
![How the ionic bond formation of sodium oxides is represented?[Hint: Atomic No. of sodium 11 , oxygen 8 ] How the ionic bond formation of sodium oxides is represented?[Hint: Atomic No. of sodium 11 , oxygen 8 ]](https://haygot.s3.amazonaws.com/questions/1758667_1836812_ans_e06a8bcc44cc427791247f1c3949bec3.png)
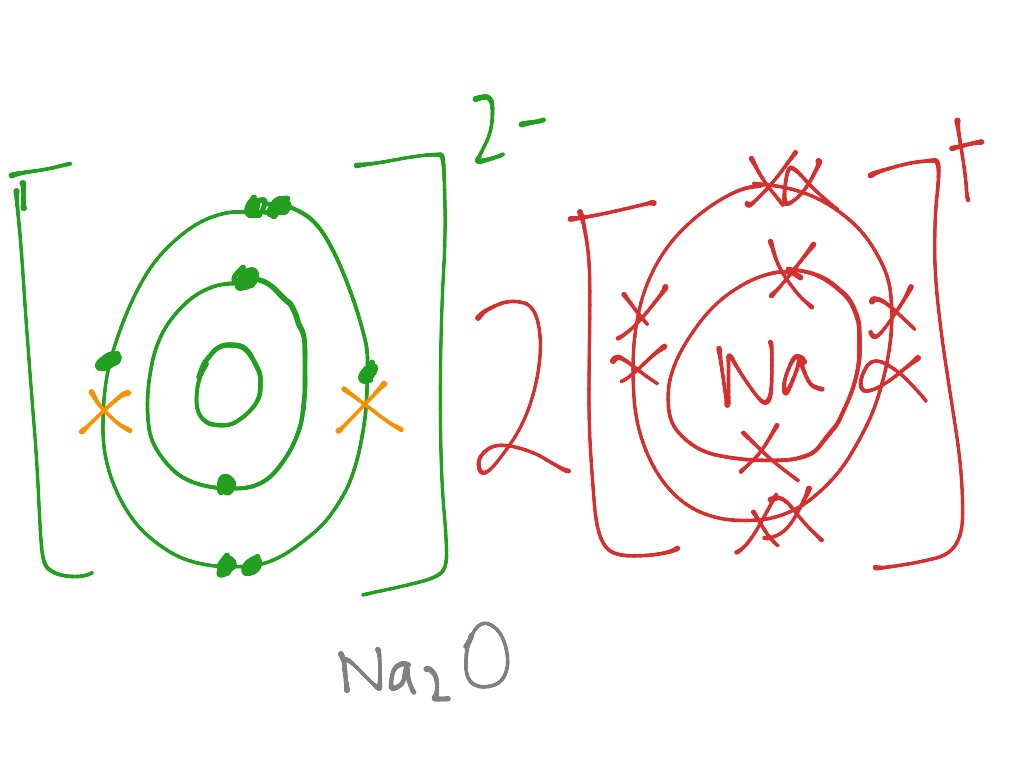
How the ionic bond formation of sodium oxides is represented?[Hint: Atomic No. of sodium 11 , oxygen 8 ]

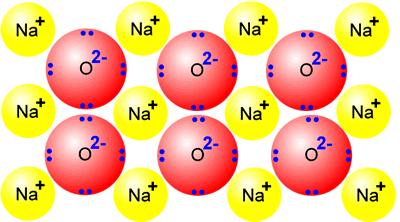
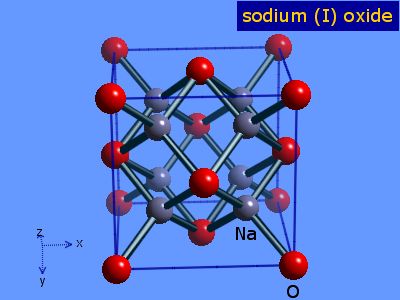
Na-Sodium Na2O- Sodium Oxide and NaCl- Sodium Chloride At standard condition- Solid Bonding and Structure: Ionic lattice Electrical Conductivity: Good. - ppt video online download