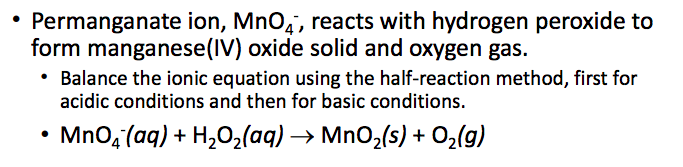
3:16 practical: investigate the effect of different solids on the catalytic decomposition of hydrogen peroxide solution - TutorMyself Chemistry

when manganese dioxide is added to hydrogen peroxide what do you obsereve - Chemistry - - 11384421 | Meritnation.com

SOLVED: 1.) Manganese (IV) Oxide acts as a catalyst in this reaction, which means it is not used up and does not appear in the products.Write out the balanced equation for the
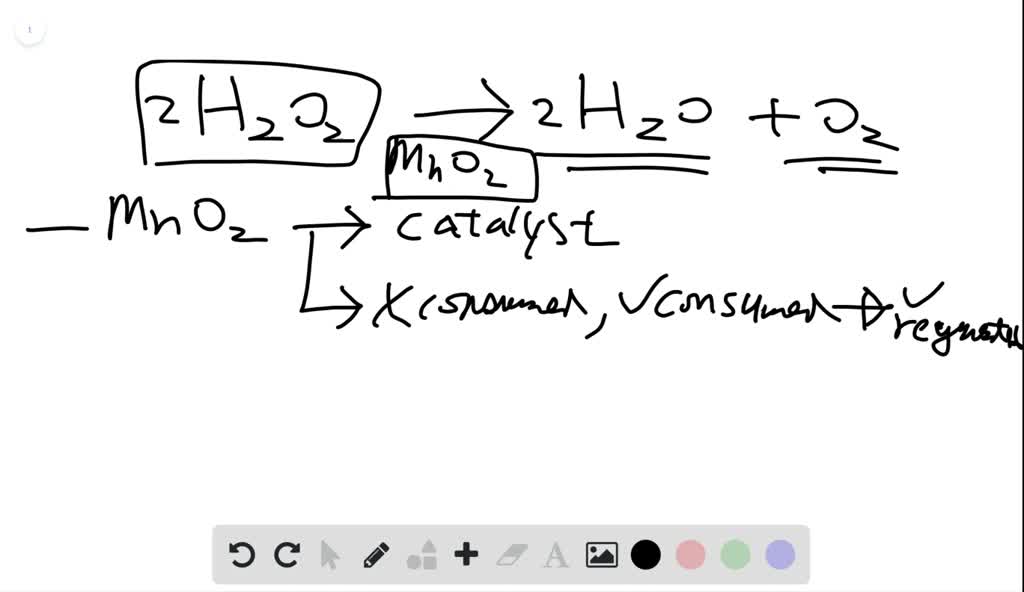
SOLVED: What happened when the manganese (IV) oxide; MnOz was added to the peroxide solution? Why did this happen? The reaction became faster because MnOz is very reactive The reaction became faster
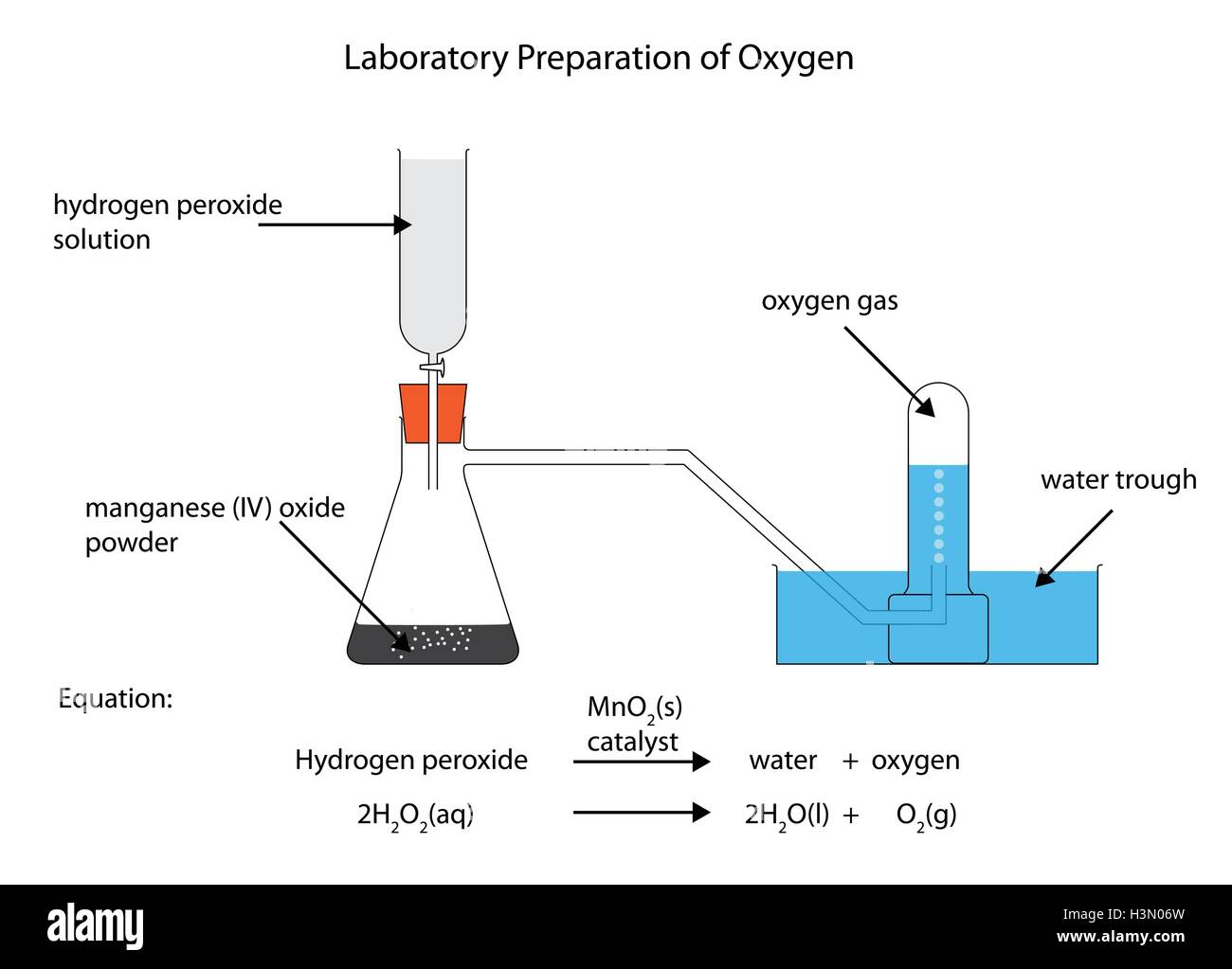
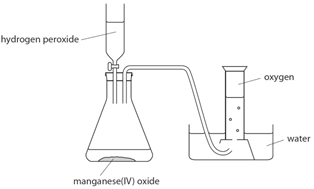
Full labelled diagram of the laboratory preparation of oxygen from hydrogen peroxide and manganese (IV) oxide Stock Vector Image & Art - Alamy

inorganic chemistry - Reaction intermediates of MnO2 catalyzed H2O2 decomposition reaction - Chemistry Stack Exchange
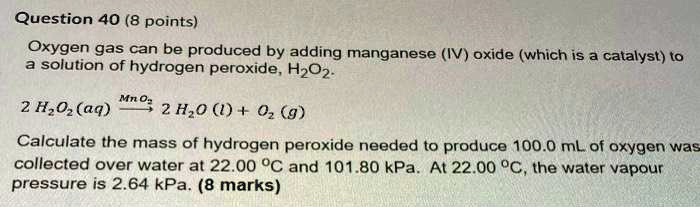
SOLVED: Question 40 (8 points) Oxygen gas can be produced by adding manganese (IV) oxide (which is a catalyst) to solution of hydrogen peroxide, H202 2 HzOz (aq) Mno 2 Hz0 (Q) +
The curves shown below were obtained when two equal volumes of hydrogen peroxide of the same - Tutorke

Question Video: Identifying the Correct Statement For the Decomposition of Hydrogen Peroxide Using a Manganese Dioxide Catalyst | Nagwa

SOLVED:A common demonstration in chemistry courses involves adding a tiny speck of manganese(IV) oxide to a concentrated hydrogen peroxide, H2 O2, solution. Hydrogen peroxide is unstable, and it decomposes quite spectacularly under

Permanganate Reduction by Hydrogen Peroxide: Formation of Reactive Manganese Species and Superoxide and Enhanced Micropollutant Abatement | ACS ES&T Engineering
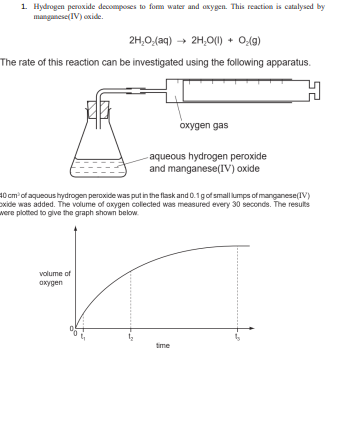
Monitoring the rate of production of oxygen from hydrogen peroxide using manganese dioxide as a catalyst


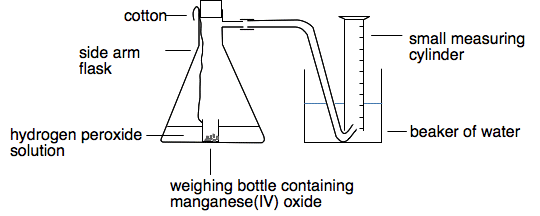
![ANSWERED] 1. What is the role of the manganese (IV... - Inorganic Chemistry ANSWERED] 1. What is the role of the manganese (IV... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/70281049-1657465695.2468812.jpeg)