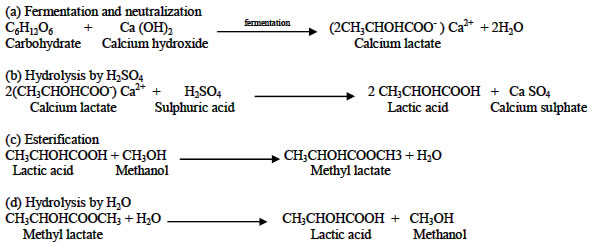
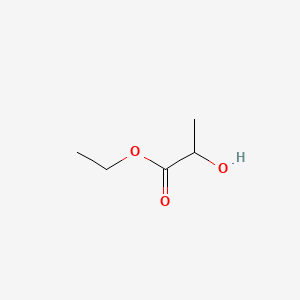
Molecules | Free Full-Text | Transesterification of Lactic Acid Oligomers with Ethanol, a Way to Anhydrous Ethyl Lactate: A Kinetic Study

Esterification of (-) - lactic acid with methanol yields (+) - methyl lactate. Assuming that there are no side reactions, what is true about this reaction?

Pervaporation Reactor for Enhanced Esterification of Lactic Acid and Isobutyl Alcohol - Chandane - 2020 - Chemical Engineering & Technology - Wiley Online Library

Molecules | Free Full-Text | Transesterification of Lactic Acid Oligomers with Ethanol, a Way to Anhydrous Ethyl Lactate: A Kinetic Study
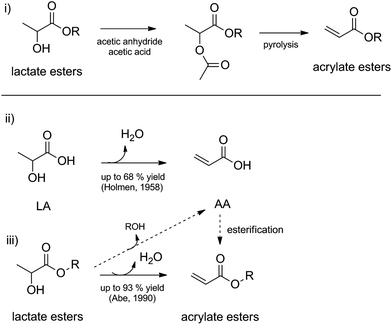
Lactic acid as a platform chemical in the biobased economy: the role of chemocatalysis - Energy & Environmental Science (RSC Publishing) DOI:10.1039/C3EE00069A

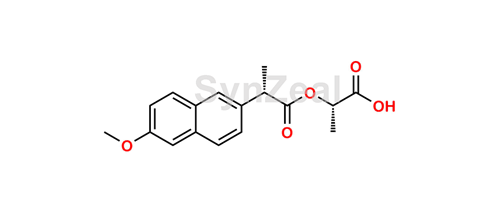
DL-Indole-3-lactic Acid Methyl Ester-D5 | 3-(3-Indolyl)-2-hydroxypropionic Acid-D5; Methyl Indole-3-lactate-D5; α-Hydroxy-1H-indole-3-propanoic Acid Methyl Ester-D5; | C₁₂H₈D₅NO₃ | TRC

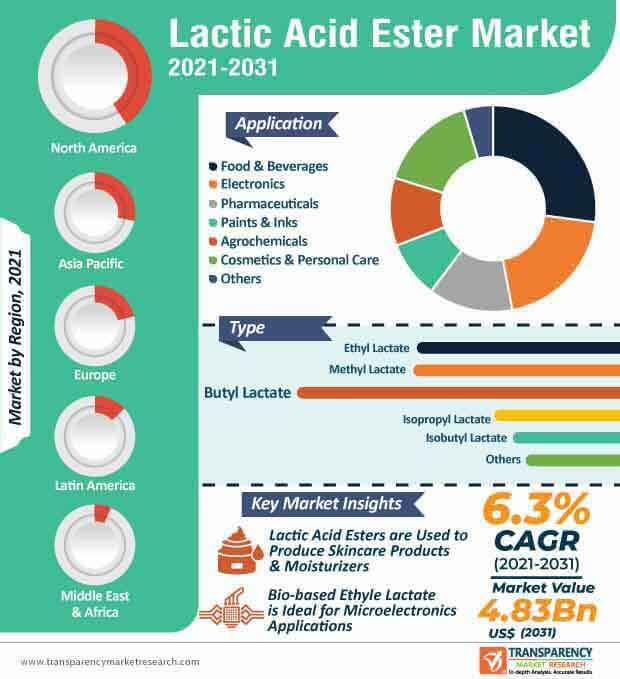
Lactic Acid Ethyl Ester Market Research Report: Key Trends and Opportunities by State and Territory for period 2022 to 2028. - MarketWatch

Use of lipases for the kinetic resolution of lactic acid esters in heptane or in a solvent free system - ScienceDirect

Esterification of Lactic Acid by Catalytic Extractive Reaction: An Efficient Way to Produce a Biosolvent Composition | SpringerLink
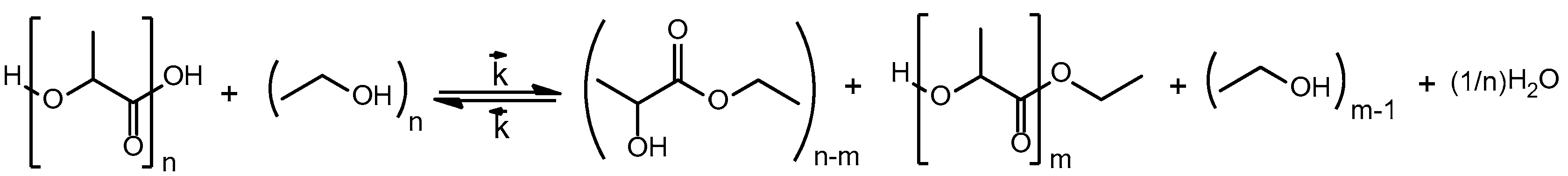
Molecules | Free Full-Text | Transesterification of Lactic Acid Oligomers with Ethanol, a Way to Anhydrous Ethyl Lactate: A Kinetic Study