![A shortcut to tris[2-(4-hydroxyphenyl)ethyl]phosphine oxide and 2-(4-hydroxyphenyl)ethylphosphinic acid via reaction of elemental phosphorus with 4-tert-butoxystyrene - ScienceDirect A shortcut to tris[2-(4-hydroxyphenyl)ethyl]phosphine oxide and 2-(4-hydroxyphenyl)ethylphosphinic acid via reaction of elemental phosphorus with 4-tert-butoxystyrene - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0959943613002125-fx1.jpg)
A shortcut to tris[2-(4-hydroxyphenyl)ethyl]phosphine oxide and 2-(4-hydroxyphenyl)ethylphosphinic acid via reaction of elemental phosphorus with 4-tert-butoxystyrene - ScienceDirect

Synthesis of phenylalanine-derived β-hydroxy and β-keto phosphine oxides — investigation of the configurational stability of lithiated phosphine oxides using the hoffmann test - ScienceDirect

Enantioselective Mukaiyama-Michael Reaction of Cyclic α-Alkylidene β-Keto Phosphine Oxide and Phosphonate and Asymmetric Synthesis of (R)-Homosarkomycin. | Semantic Scholar
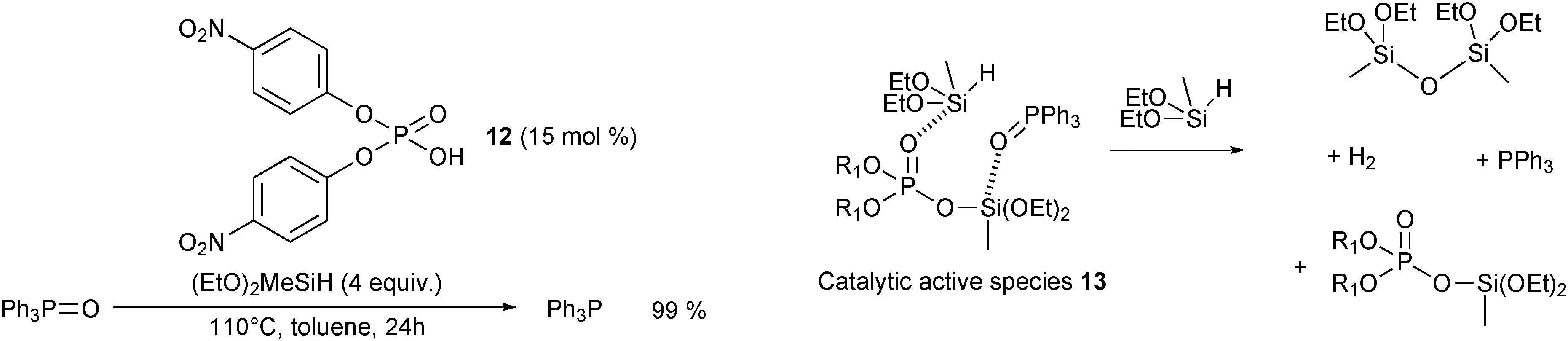
Reduction of secondary and tertiary phosphine oxides to phosphines - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C4CS00311J

Enzyme-Inspired Chiral Secondary-Phosphine-Oxide Ligand with Dual Noncovalent Interactions for Asymmetric Hydrogenation. | Semantic Scholar
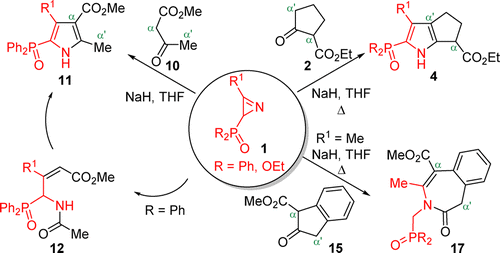
Reaction of 2H-Azirine-Phosphine Oxides and -Phosphonates with Enolates Derived from β-Keto Esters,The Journal of Organic Chemistry - X-MOL

MnIII-catalyzed phosphorylation of vinyl azides: The synthesis of β-keto phosphine oxides - ScienceDirect

General and Selective Copper-Catalyzed Reduction of Tertiary and Secondary Phosphine Oxides: Convenient Synthesis of Phosphines | Journal of the American Chemical Society
When aldehydes/ketones are treated with phosphorus Yilid, what type of hydrocarbon is produced? Explain its reaction mechanism. - Quora
![Oxidation of an Internal‐Edge‐Substituted [5]Helicene‐Derived Phosphine Synchronously Enhances Circularly Polarized Luminescence - Usui - 2022 - Chemistry – A European Journal - Wiley Online Library Oxidation of an Internal‐Edge‐Substituted [5]Helicene‐Derived Phosphine Synchronously Enhances Circularly Polarized Luminescence - Usui - 2022 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/b9b91d61-ac97-4c90-8342-ba4b1673af5b/chem202202922-toc-0001-m.jpg)
Oxidation of an Internal‐Edge‐Substituted [5]Helicene‐Derived Phosphine Synchronously Enhances Circularly Polarized Luminescence - Usui - 2022 - Chemistry – A European Journal - Wiley Online Library

Enzyme-Inspired Chiral Secondary-Phosphine-Oxide Ligand with Dual Noncovalent Interactions for Asymmetric Hydrogenation. | Semantic Scholar

N‐Phosphine Oxide‐Substituted Imidazolylidenes (PoxIms): Multifunctional Multipurpose Carbenes - Hazra - 2017 - Chemistry – A European Journal - Wiley Online Library
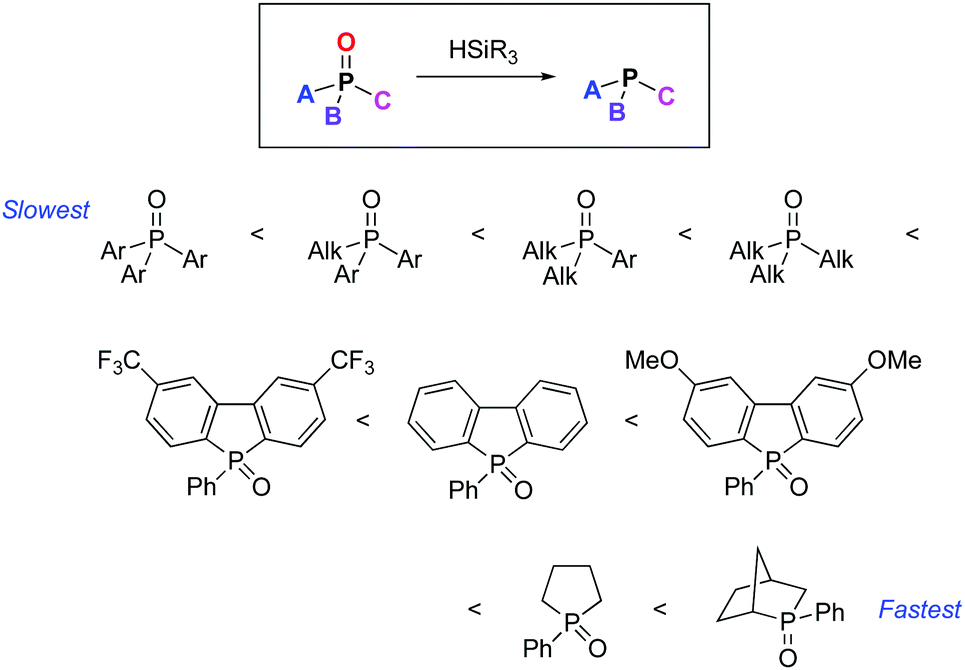
Why do silanes reduce electron-rich phosphine oxides faster than electron-poor phosphine oxides? - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC08718D

Catalyst- and Solvent-Free Hydrophosphorylation of Ketones with Secondary Phosphine Oxides: Green Synthesis of Tertiary α-Hydroxyphosphine Oxides
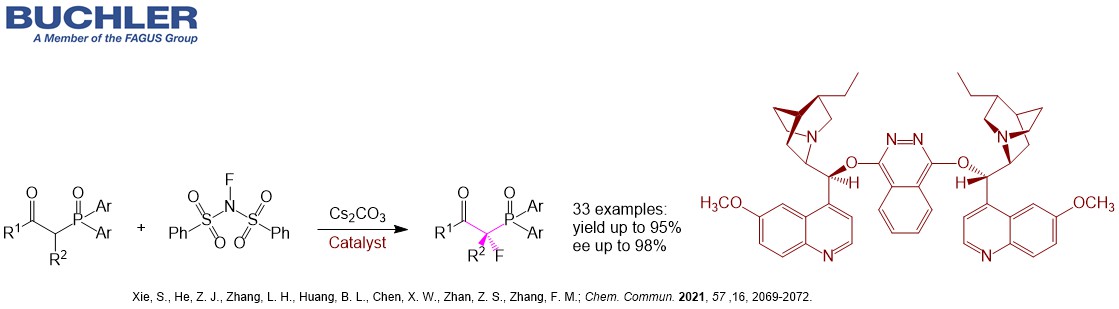
Fluorination, beta-ketodiaryl-phosphine oxides, N-fluoro-N-(phenylsulfonyl)benzenesulfonamide, fluorinated beta-ketodiaryl-phosphine oxides, 1,4-Bis(9-O-dihydroquinidinyl)phthalazine, Dihydroquinidine Derivative - Buchler GmbH