Succinimide formation at Asn 55 in the complementarity determining region of a recombinant monoclonal antibody IgG1 heavy chain. | Semantic Scholar
Minimizing Method-Induced Deamidation and Isomerization During Antibody Characterization to Ensure Optimal Understanding of Product Quality Attributes
![PDF] Aspartic Acid Isomerization Characterized by High Definition Mass Spectrometry Significantly Alters the Bioactivity of a Novel Toxin from Poecilotheria | Semantic Scholar PDF] Aspartic Acid Isomerization Characterized by High Definition Mass Spectrometry Significantly Alters the Bioactivity of a Novel Toxin from Poecilotheria | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a4a6100df8b6ec88f29727dc5e0fae91a2d67fce/3-Figure1-1.png)
PDF] Aspartic Acid Isomerization Characterized by High Definition Mass Spectrometry Significantly Alters the Bioactivity of a Novel Toxin from Poecilotheria | Semantic Scholar

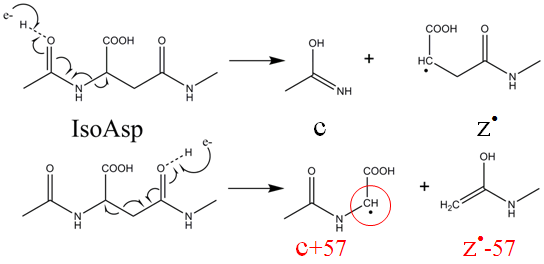
Deamidation: Differentiation of aspartyl from isoaspartyl products in peptides by electron capture dissociation - Cournoyer - 2005 - Protein Science - Wiley Online Library
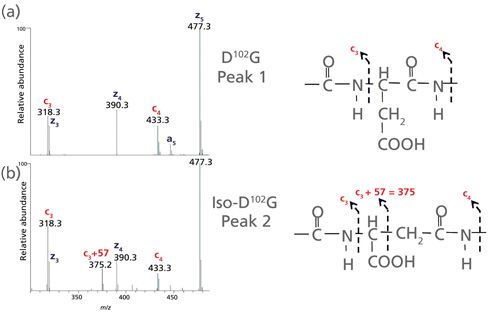
Distinguishing Aspartic and Isoaspartic Acids in Peptides by Several Mass Spectrometric Fragmentation Methods | SpringerLink
Distinguishing d - and l -aspartic and isoaspartic acids in amyloid β peptides with ultrahigh resolution ion mobility spectrometry - Chemical Communications (RSC Publishing) DOI:10.1039/C7CC03321D

Toxins | Free Full-Text | Aspartic Acid Isomerization Characterized by High Definition Mass Spectrometry Significantly Alters the Bioactivity of a Novel Toxin from Poecilotheria

High-Resolution Demultiplexing (HRdm) Ion Mobility Spectrometry–Mass Spectrometry for Aspartic and Isoaspartic Acid Determination and Screening | Analytical Chemistry

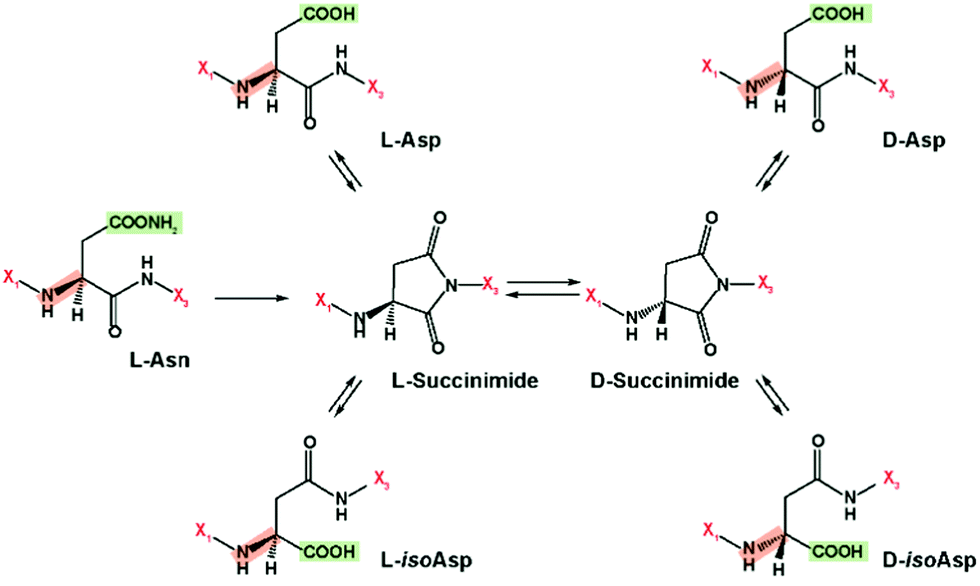
Schematic of asparagine deamidation via succinimide intermediate to... | Download Scientific Diagram
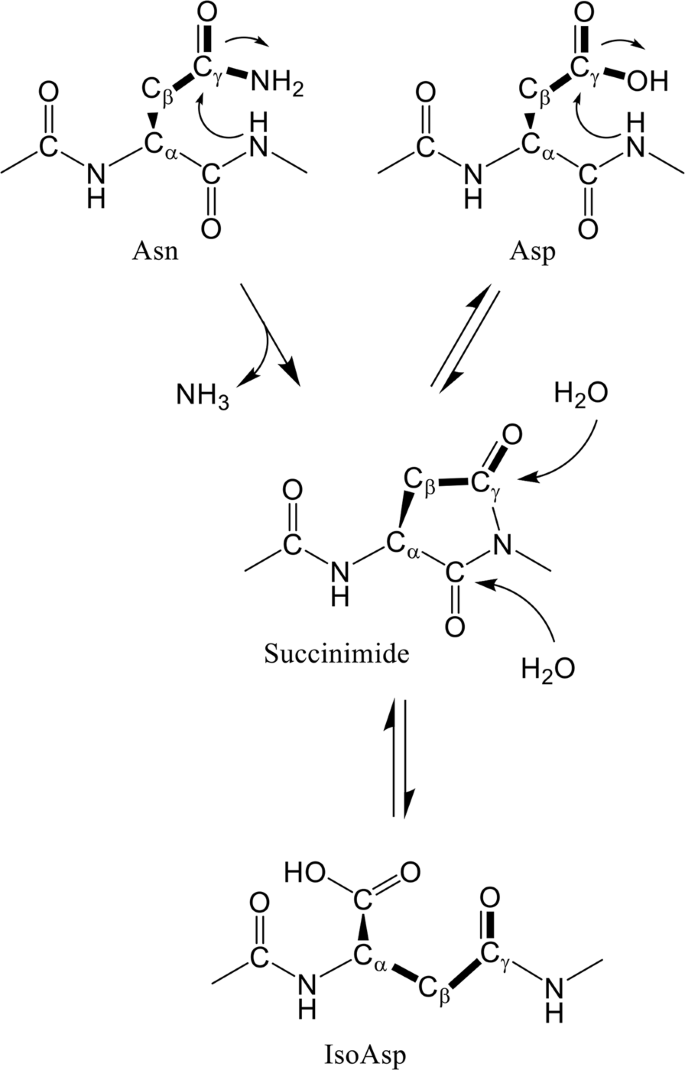
Structural and biochemical basis of the formation of isoaspartate in the complementarity-determining region of antibody 64M-5 Fab | Scientific Reports
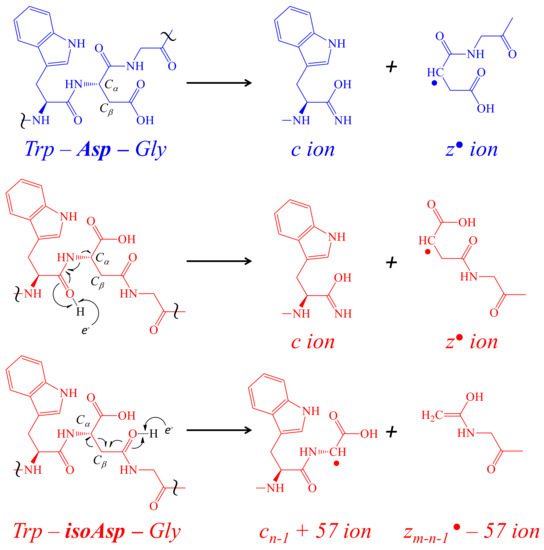
Toxins | Free Full-Text | Aspartic Acid Isomerization Characterized by High Definition Mass Spectrometry Significantly Alters the Bioactivity of a Novel Toxin from Poecilotheria

Independent highly sensitive characterization of asparagine deamidation and aspartic acid isomerization by sheathless CZE‐ESI‐MS/MS - Gahoual - 2016 - Journal of Mass Spectrometry - Wiley Online Library

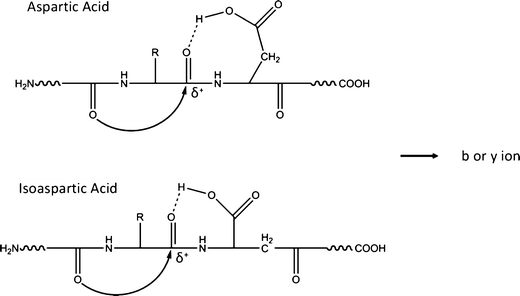
Electron Transfer Dissociation with Supplemental Activation to Differentiate Aspartic and Isoaspartic Residues in Doubly Charged Peptide Cations - ScienceDirect

PIMT-Mediated Labeling of l-Isoaspartic Acid with Tris Facilitates Identification of Isomerization Sites in Long-Lived Proteins | Journal of the American Society for Mass Spectrometry

Frontiers | Identification, Efficacy, and Stability Evaluation of Succinimide Modification With a High Abundance in the Framework Region of Golimumab