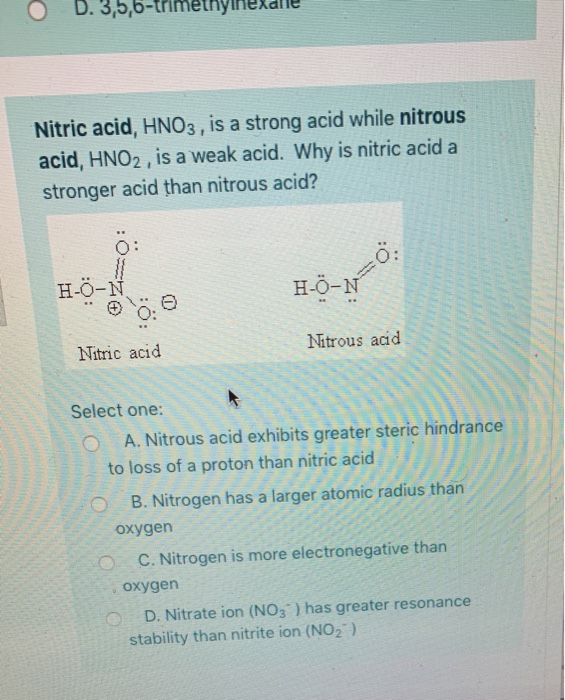
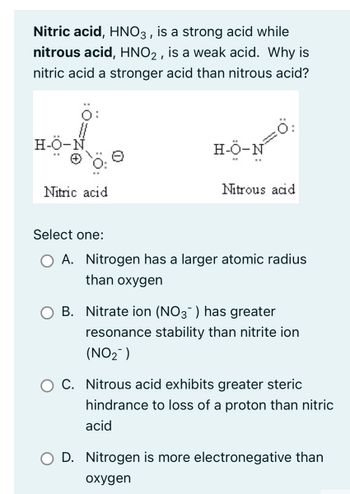
Statement I : HNO3 is a stronger acid than HNO2. - Sarthaks eConnect | Largest Online Education Community

SOLVED: 10. Explain - the following = observations: H2Se is a 'stronger acid than H2S I.H-Se '5 Iess Qolr #n h-S 2,04-k '5 ^kh sjv # HS 3.Hse crbatel ovvky iItsc In

Nitric acid, HNO2 is a weak acid with Ka = 4.5 x 10-4. It dissociates according to: HNO2 (aq) - Home Work Help - Learn CBSE Forum
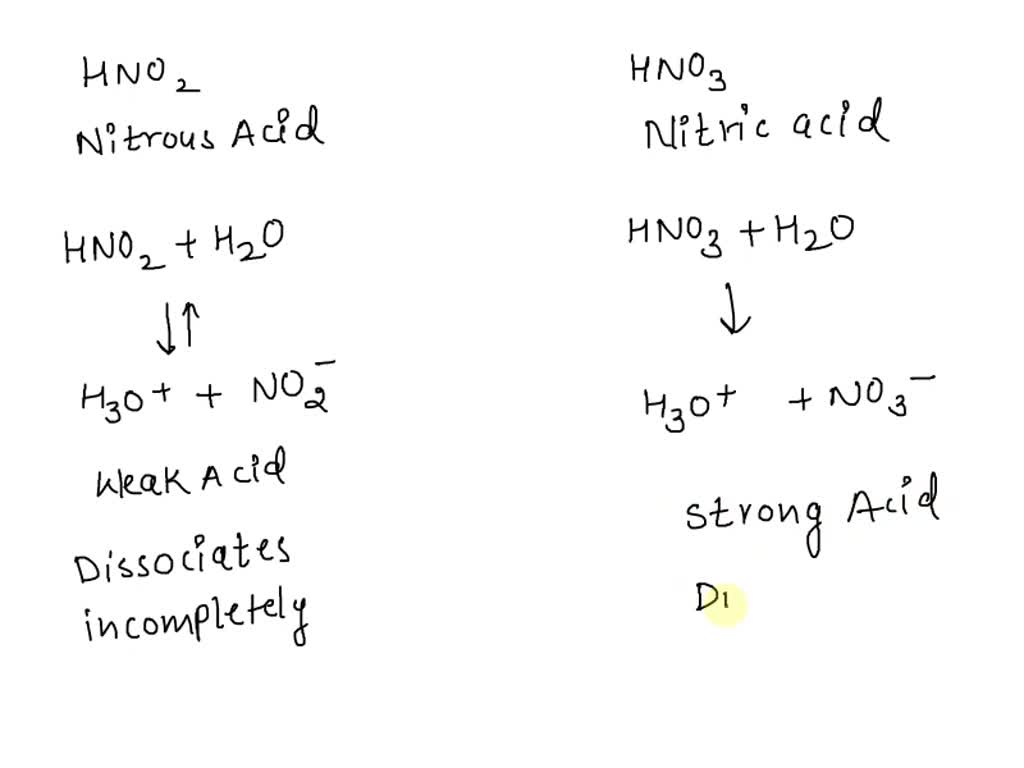
SOLVED: A conductivity test of a dilute solution of nitrous acid (HNO2(aq)) has very low electrical conductivity (the bulb on the tester glows dimly) and the pH is measured to be about