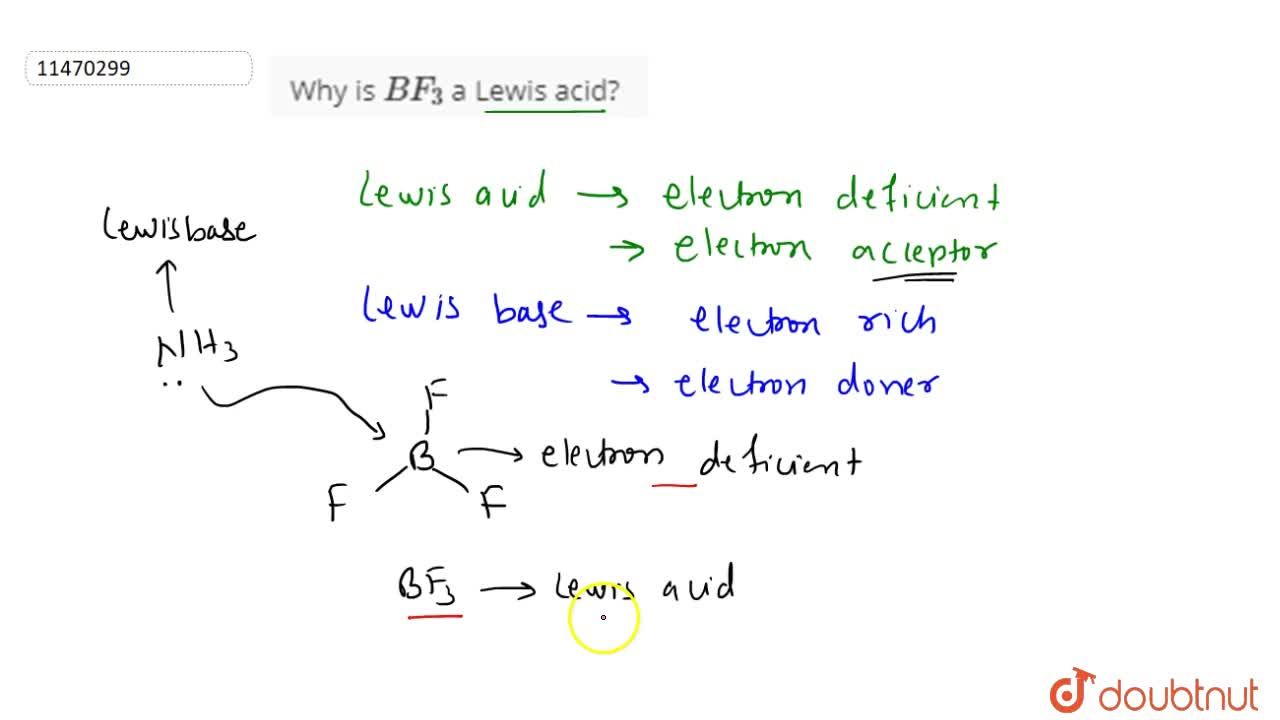
Why BF3 acts as Lewis Acid | Boron trifluoride acts as a Lewis Acid why | Lewis Acid and Base Theory - YouTube

Draw the product of the following Lewis acid-base reaction below. Label the electrophile and nucleophile. | Homework.Study.com

Classify the Substances as Lewis Acids or Lewis Bases | Identifying Lewis Acids and Bases Practice - YouTube
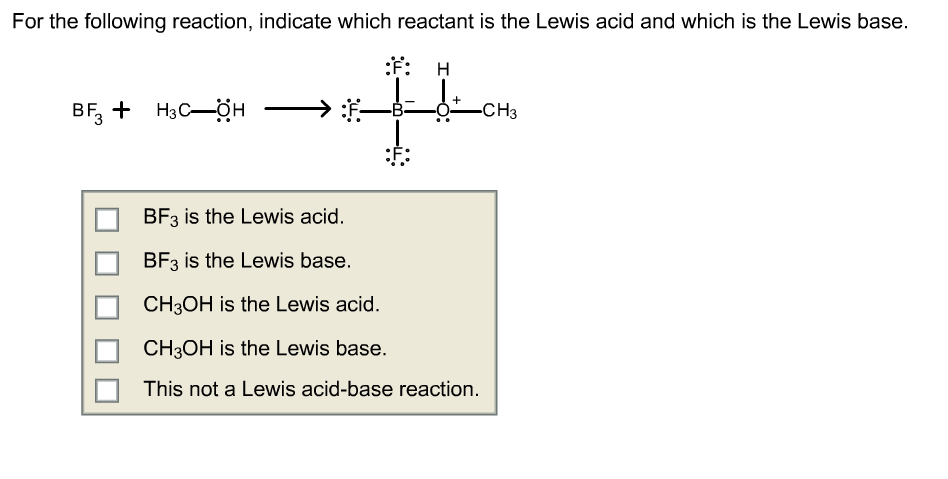
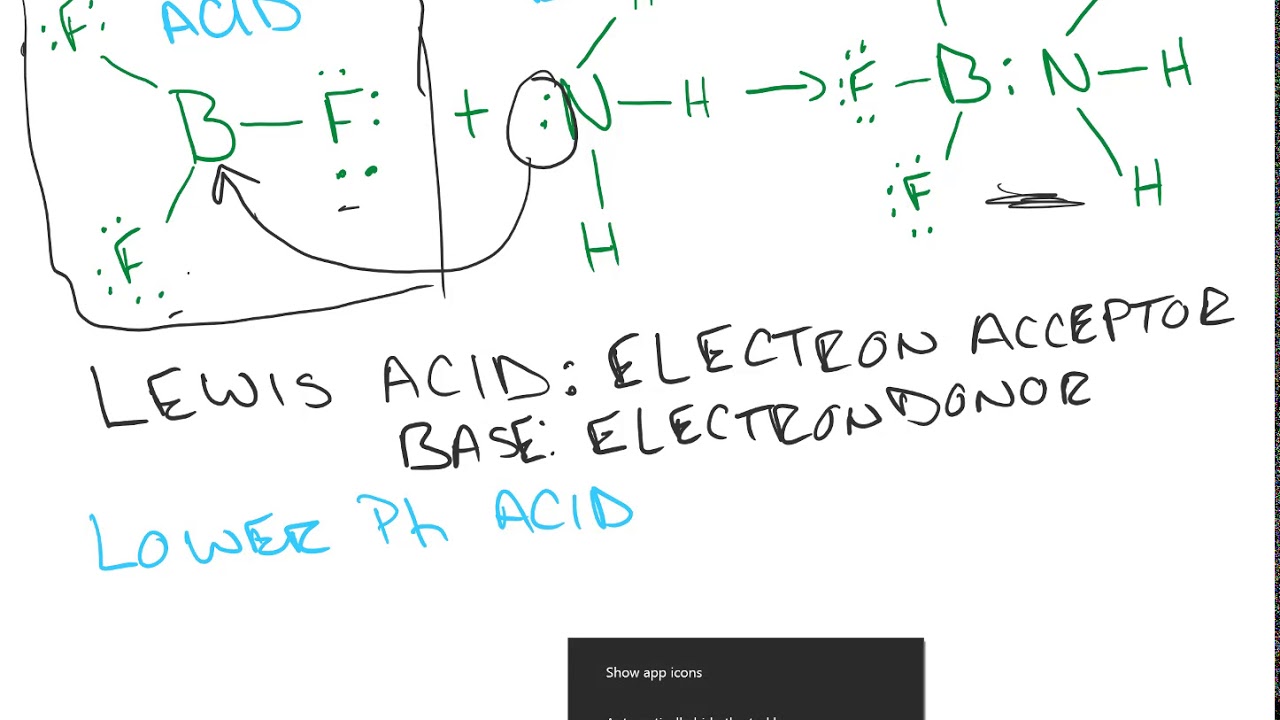
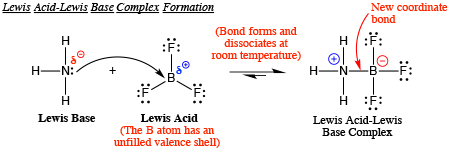
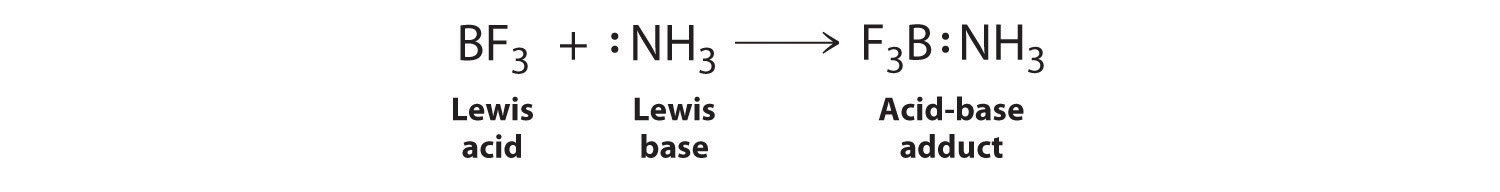
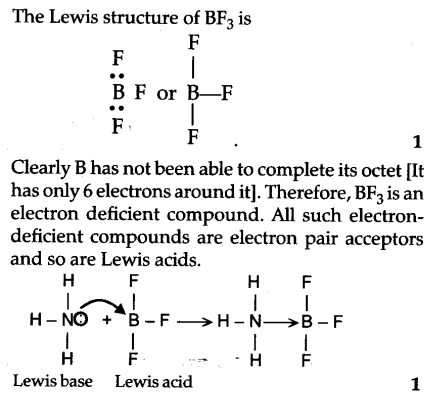
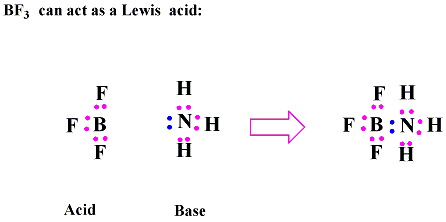
Explain : (A) BF3 is a Lewis acid, (B) NH3 is a Lewis base. - Sarthaks eConnect | Largest Online Education Community
Why does boron trifluoride (BF3) act as a Lewis base and ammonia (NH3) acts as a Lewis acid? - Quora

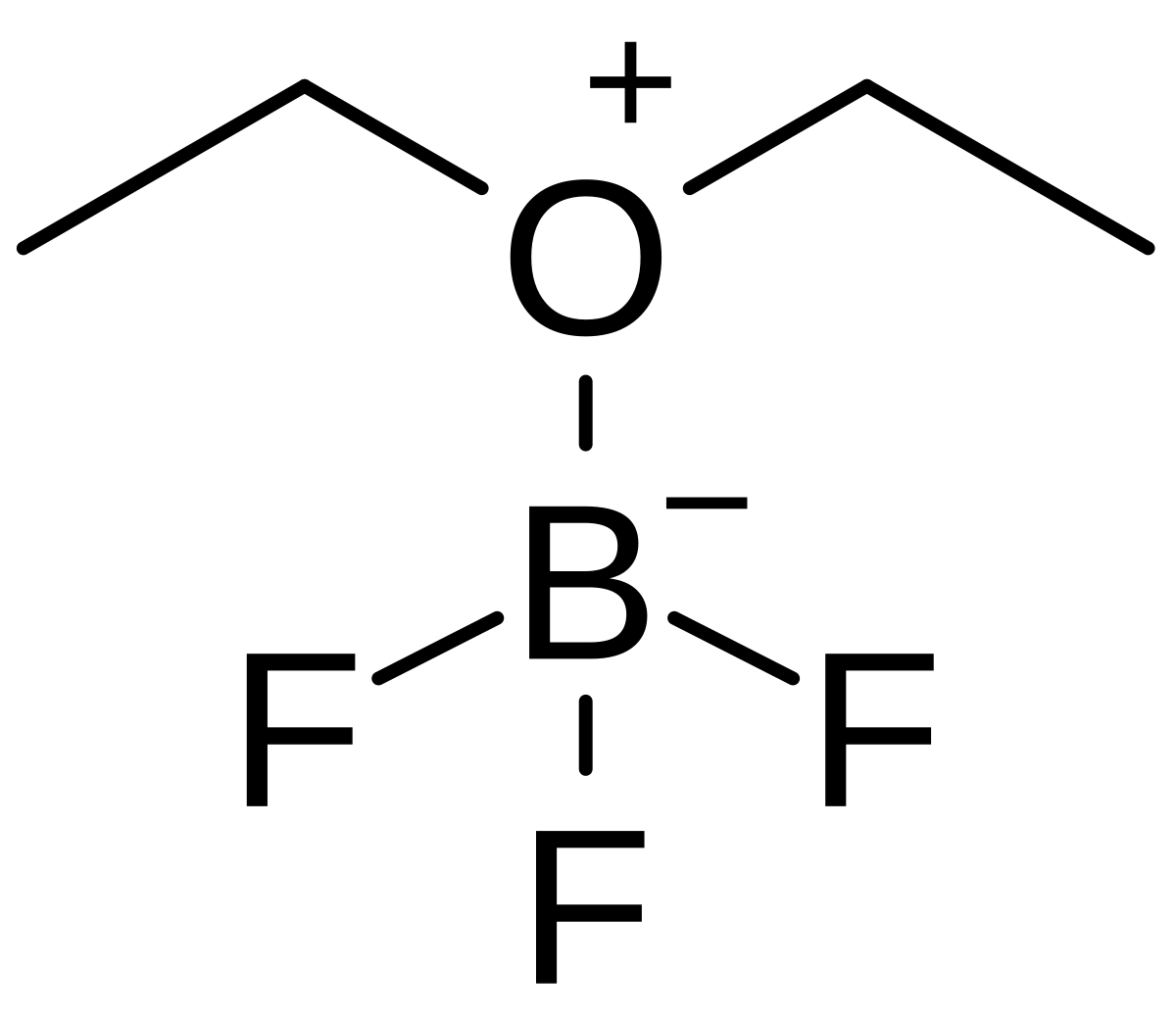
Boron trifluoride, BF3, and ammonia, NH3, react to produce BF3 : NH3. A coordinate covalent bond is formed between the boron atom on BF3 and the nitrogen atom on NH3. Write the
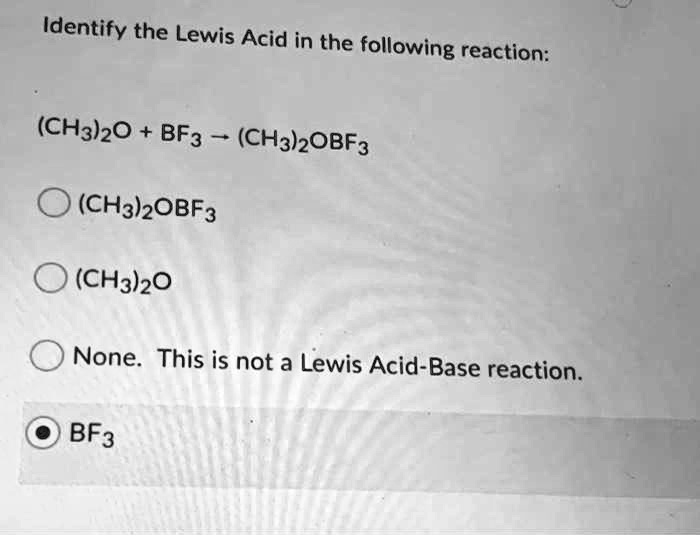
SOLVED: Identify the Lewis Acid in the following reaction: (CH3)2o BF3 (CH3)OBF3 (CH3)OBF3 (CH3h)20 None This is not a Lewis Acid-Base reaction. BF3

Question Video: Identifying the Species That Is Not a Lewis Acid in a Set of Chemical Formulas | Nagwa