OneClass: Write a balanced equation for the reaction that occurs wher (a) iron(III) oxide is reduced ...
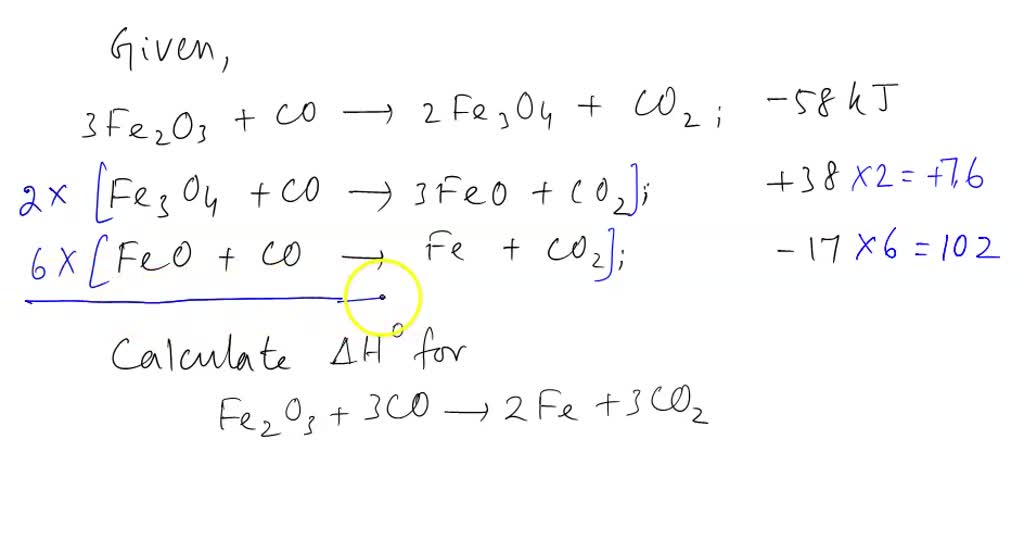
SOLVED: Part 1) Use the following equations to calculate the heat of the reaction for the reduction of iron oxide to iron with carbon monoxide. 3Fe2O3(s) + CO(g) → 2Fe3O4(s) + CO2(g)

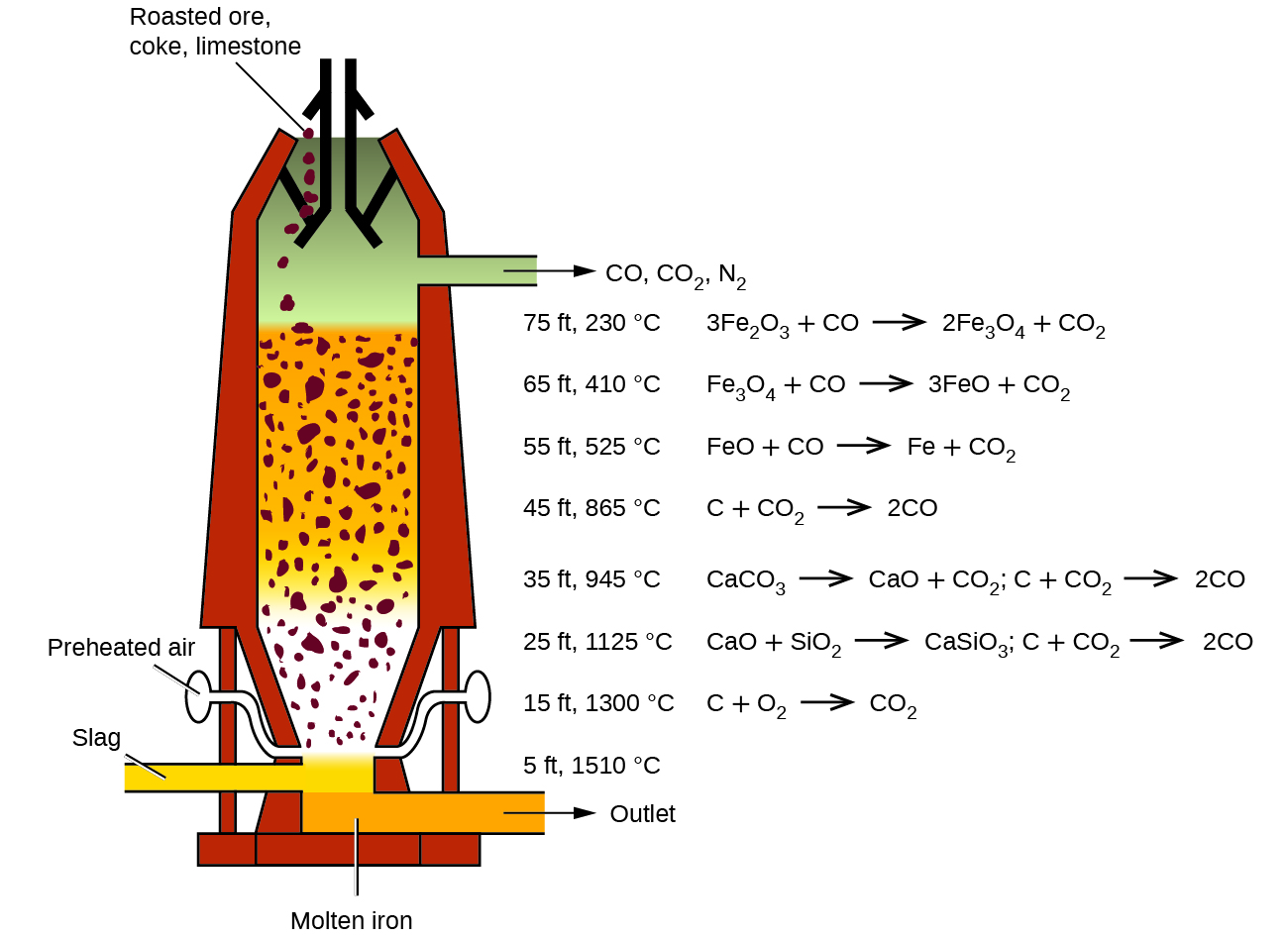
Question Video: Identifying the Compound That Reduces Iron Ore inside a Working Blast Furnace | Nagwa

PDF) Influence of hydrogen and various carbon monoxide concentrations on reduction behavior of iron oxide at low temperature

Iron(iii)oxide+Carbon monoxide=Iron+ Carbon dioxide Balanced Equation ||Fe2O3+CO=Fe+CO2 Balanced Equ. - YouTube

Reduction of Carbon Dioxide by a Molybdenum-Containing Formate Dehydrogenase: A Kinetic and Mechanistic Study | Journal of the American Chemical Society

SOLVED: 2 Elemental iron can be recovered from iron (III) oxide ore by reacting with carbon monoxide (UNBALANCED chemical equation below): FezO3 (s) CO (g) COz (g) Fe (s) (2 points) Balance