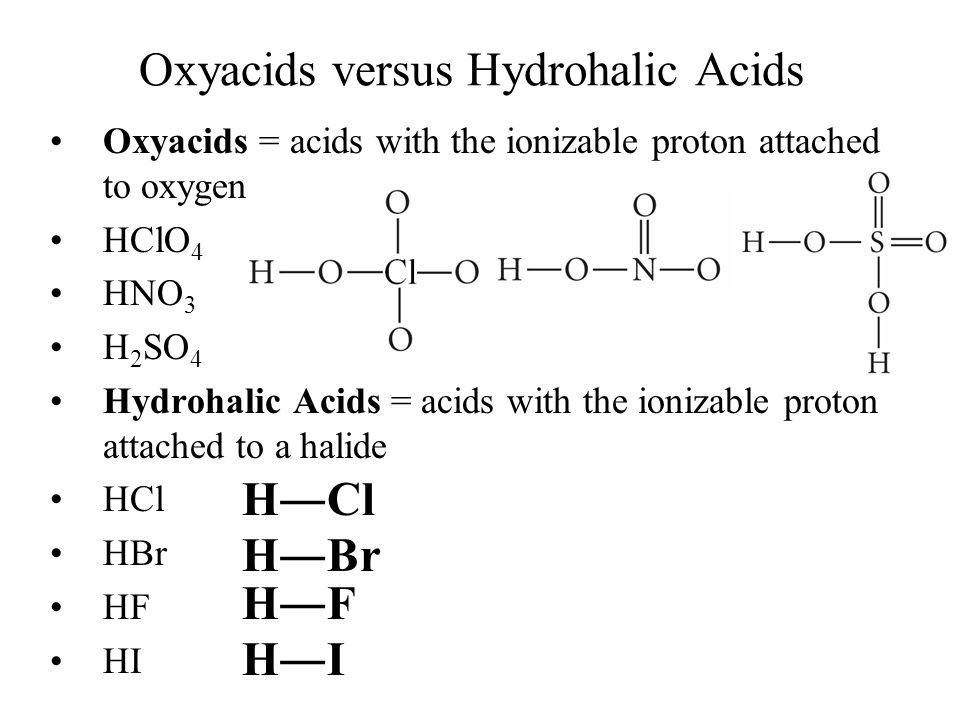
![PDF] One-pot hydrogen peroxide and hydrohalic acid induced ring closure and selective aromatic halogenation to give new ring-fused benzimidazoles. | Semantic Scholar PDF] One-pot hydrogen peroxide and hydrohalic acid induced ring closure and selective aromatic halogenation to give new ring-fused benzimidazoles. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/71ef4373e544f48c805b6e08393f373d35de21fc/2-Table1-1.png)
PDF] One-pot hydrogen peroxide and hydrohalic acid induced ring closure and selective aromatic halogenation to give new ring-fused benzimidazoles. | Semantic Scholar
![PDF] One-pot hydrogen peroxide and hydrohalic acid induced ring closure and selective aromatic halogenation to give new ring-fused benzimidazoles. | Semantic Scholar PDF] One-pot hydrogen peroxide and hydrohalic acid induced ring closure and selective aromatic halogenation to give new ring-fused benzimidazoles. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/71ef4373e544f48c805b6e08393f373d35de21fc/4-Table3-1.png)
PDF] One-pot hydrogen peroxide and hydrohalic acid induced ring closure and selective aromatic halogenation to give new ring-fused benzimidazoles. | Semantic Scholar

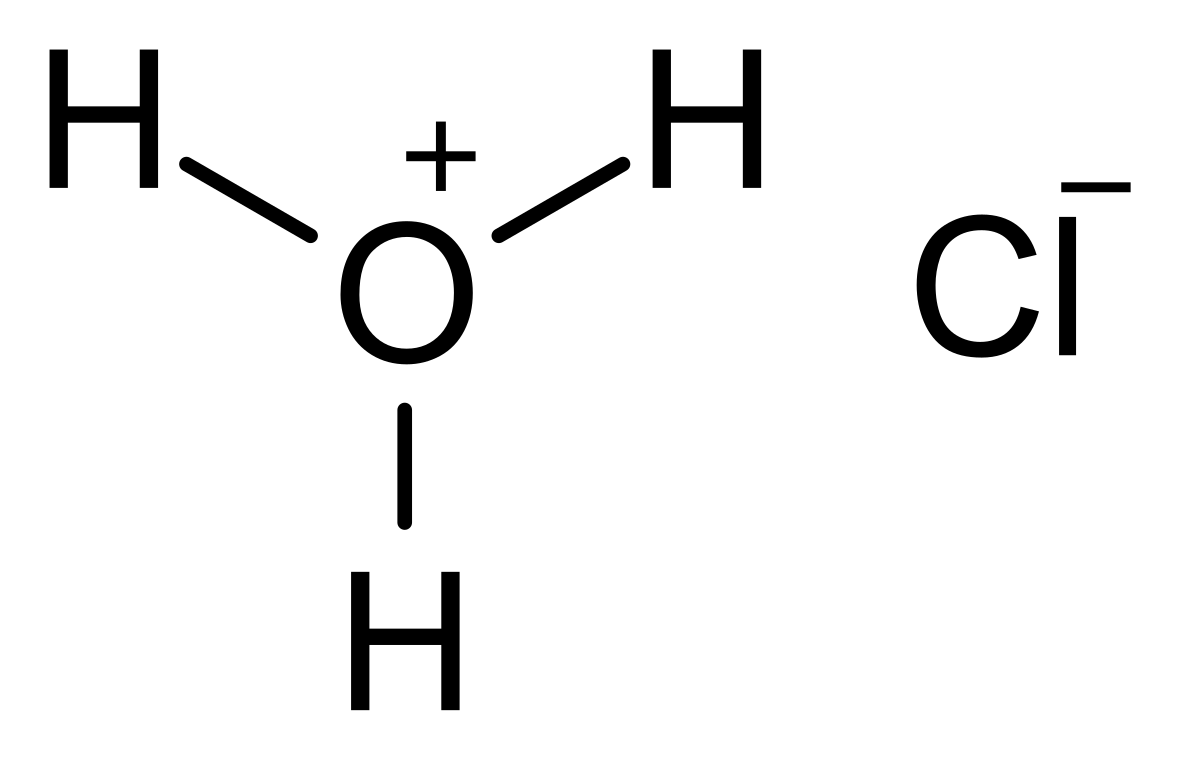
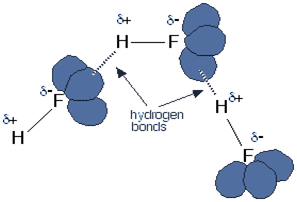
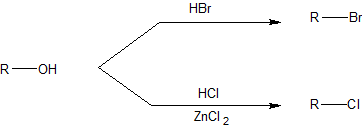
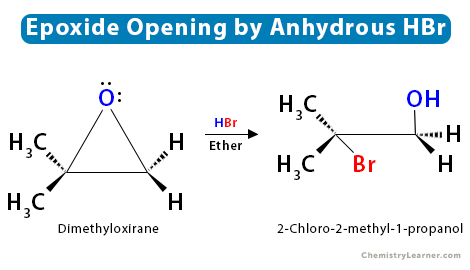
SOLVED: Rank the following acids: HCl, HI, HBr, HF according to acid strength from weakest to strongest. Explain the observed trend in hydrohalic acid strength (strength of the hydrohalic acid formed from