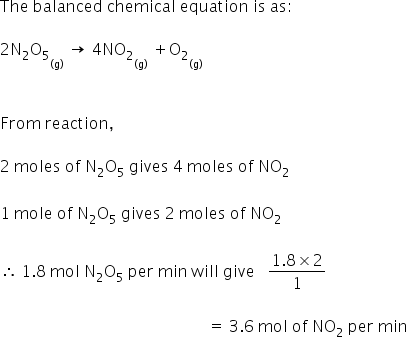Molecules | Free Full-Text | N2O Decomposition over Fe-ZSM-5: A Systematic Study in the Generation of Active Sites

Table 1 from Mechanism and kinetics of direct N2O decomposition over Fe-MFI zeolites with different iron speciation from temporal analysis of products. | Semantic Scholar

On the mechanism of high-temperature N2O decomposition over Fe-FER in the presence of NO - ScienceDirect

Recent Advances on Nitrous Oxide (N2O) Decomposition over Non-Noble-Metal Oxide Catalysts: Catalytic Performance, Mechanistic Considerations, and Surface Chemistry Aspects | ACS Catalysis
![Data for the decomposition of dinitrogen oxide on a gold surface at 900 degrees Celsius are given below. Verify that the reaction is first order by preparing a graph of ln ~[N2O] Data for the decomposition of dinitrogen oxide on a gold surface at 900 degrees Celsius are given below. Verify that the reaction is first order by preparing a graph of ln ~[N2O]](https://homework.study.com/cimages/multimages/16/at16316449417038598771.jpg)
Data for the decomposition of dinitrogen oxide on a gold surface at 900 degrees Celsius are given below. Verify that the reaction is first order by preparing a graph of ln ~[N2O]

Recent Advances on Nitrous Oxide (N2O) Decomposition over Non-Noble-Metal Oxide Catalysts: Catalytic Performance, Mechanistic Considerations, and Surface Chemistry Aspects | ACS Catalysis

SOLVED: The experimental rate law for the decomposition of nitrous oxide (NzO) to Nz and 0z is rate = kINz0J2. Two mechanisms are sproposed NzO - Nz + 0 L NzO +
✓ Solved: Sketch a potential-energy diagram for the decomposition of nitrous oxide. N2O(g)→ N2(g)+O(g)...

consider the thermal decomposition of nitrous oxide according to the equation 2N2O gives 2N2 +O2 The rate law for - Chemistry - Chemical Kinetics - 11700392 | Meritnation.com

Catalytic decomposition of nitrous oxide by gold at `900^(@)C` at an initial pressure of 200mm - YouTube
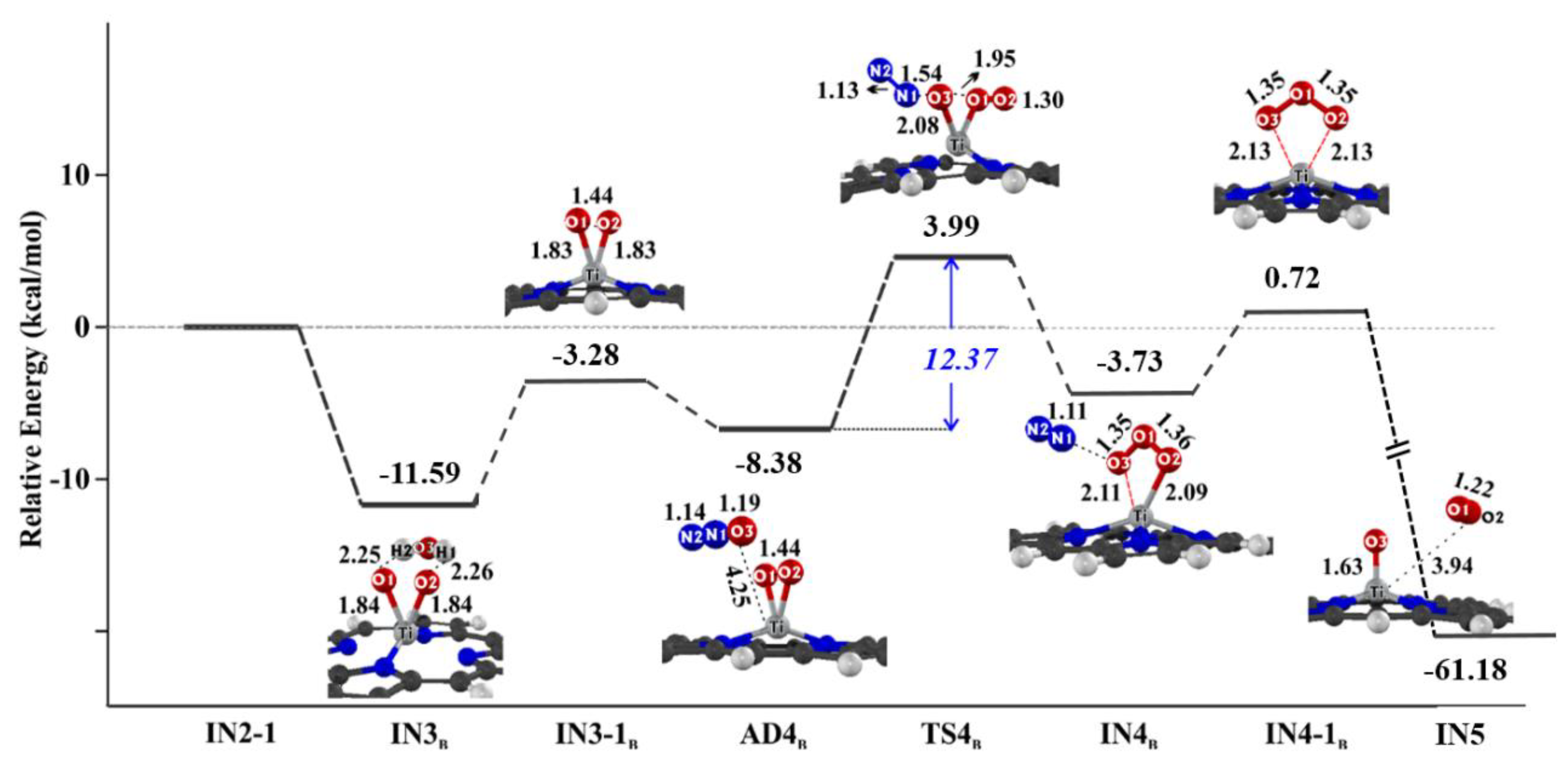
Catalysts | Free Full-Text | Effect of Water Molecule on Photo-Assisted Nitrous Oxide Decomposition over Oxotitanium Porphyrin: A Theoretical Study