
what happens when copper reacts with concentrated nitric acid give the balance chemical equation for - Brainly.in

تويتر \ Compound Interest على تويتر: "Reacting copper with concentrated nitric acid: http://t.co/CkQhEbTt5b"

When copper is treated with a certain concentration of nitric acid, nitric oxide and nitrogen dioxide are liberated in equal volumes according to the equation: xCu + yHNO3⟶ Cu (NO3)2 + NO +
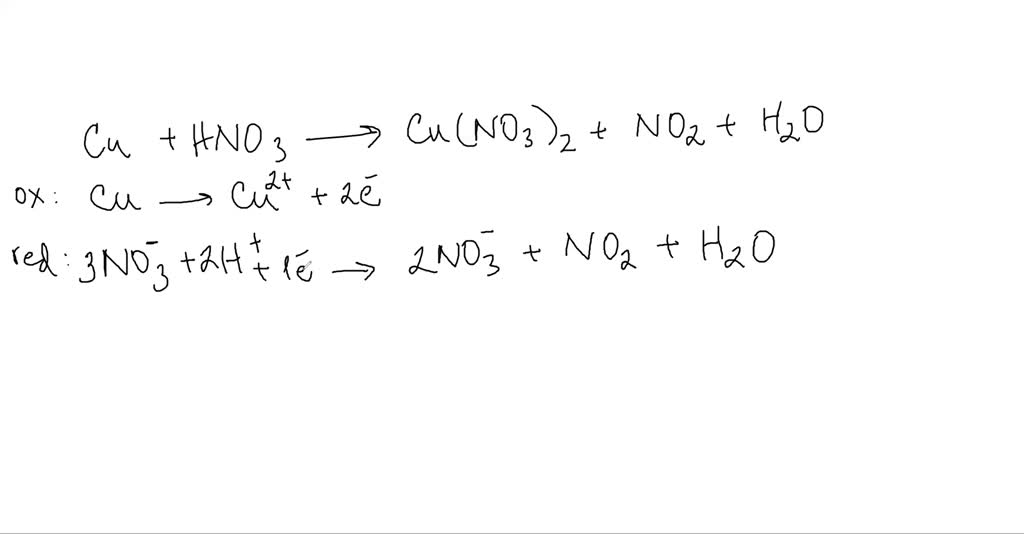
SOLVED:When solid copper is added to nitric acid, copper(II) nitrate, nitrogen dioxide, and water are produced. Write the balanced chemical equation for the reaction. List six mole ratios for the reaction.

Reaction of copper with Nitric acid (HNO3), also known as aqua fortis and spirit of niter, is a highly corrosive mineral acid Stock Photo - Alamy












2%20+%20NO%20+%20H2O%20reaction.jpg?ezimgfmt=rs:323x202/rscb1/ngcb1/notWebP)






