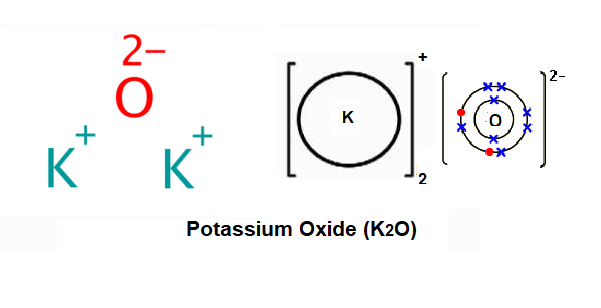
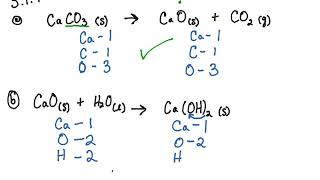Solid calcium oxide was taken in a container and water was added slowly to it. (i) State tw - YouTube

SOLVED: Ammonia gas can be prepared by the reaction of a metal oxide such as calcium oxide with ammonium chloride: CaO(s) + 2 NH4 Cl(s) 72 NH (g) + HzO(g) + Ca

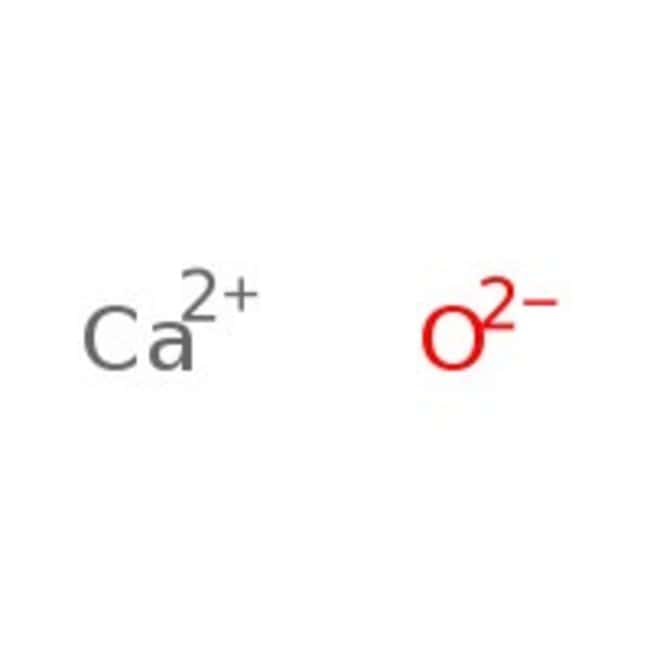
SOLVED: Caldum oxide reacts with water in combination reaction produce calcium hydroxide; CaO (s) H2O (I) Ca(OH)2 (s) 3.50-8 sample of CaO is reacted with 3.38 gof HzO How Many grams of