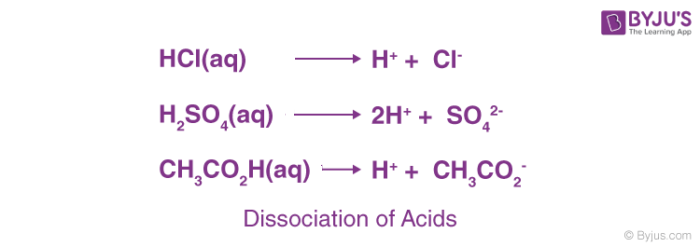
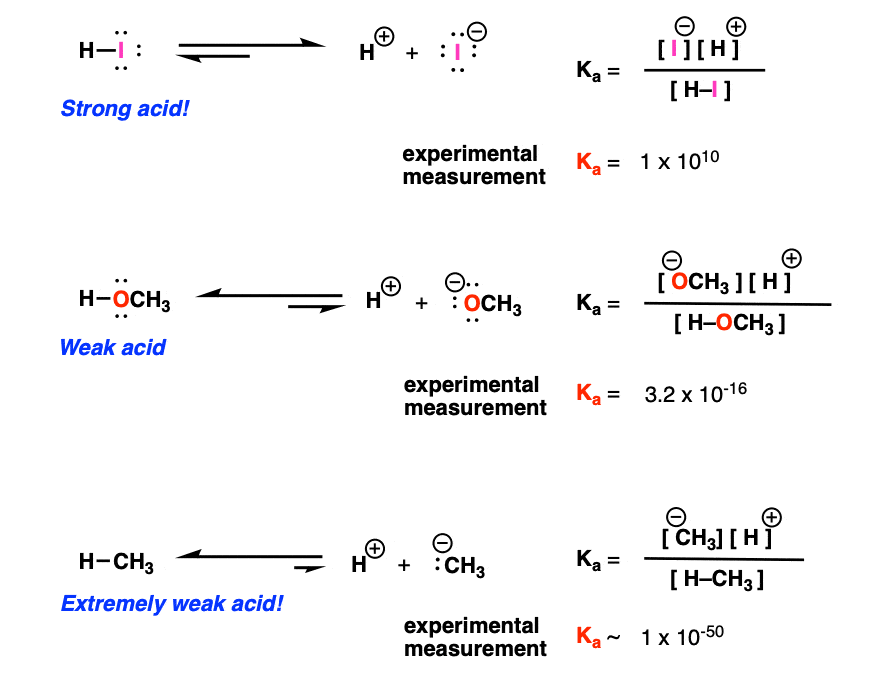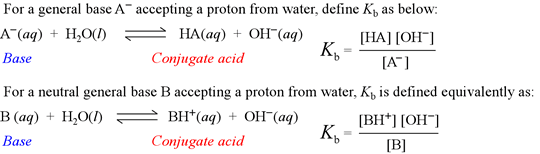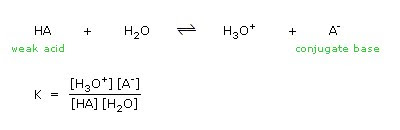Ionization of Acid And Bases - Arrhenius concept of Acid And Base Ionisation, Explanation, Determination ionisation constant of Acid base, Examples And FAQS

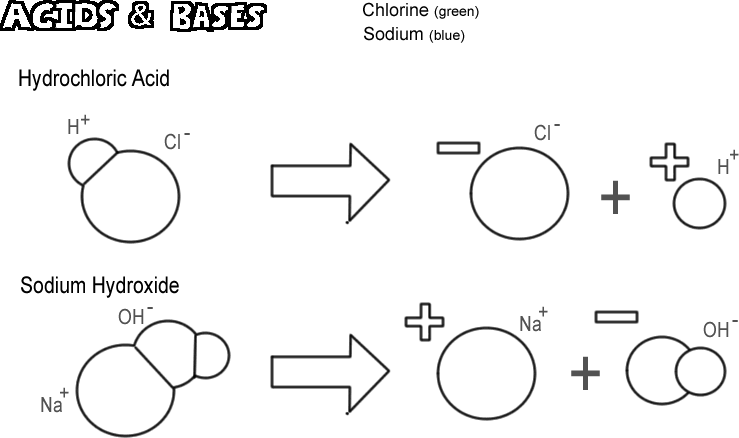
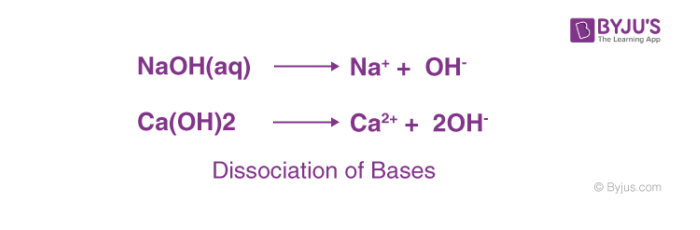
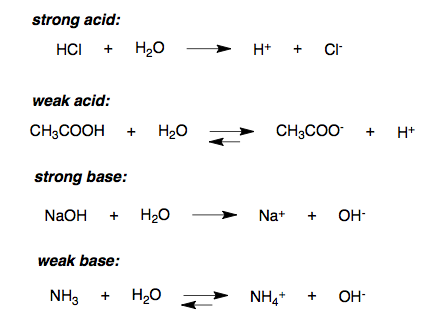
A- Label the reactions as weak acid dissociation , strong acid, weak base, strong base dissociation, or - Brainly.com

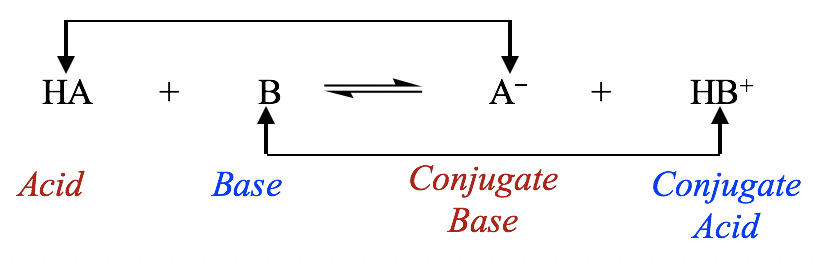
ACIDS AND BASES Dissociation Constants. weaker the acid, the stronger its conjugate base stronger the acid, the weaker its conjugate base. - ppt download